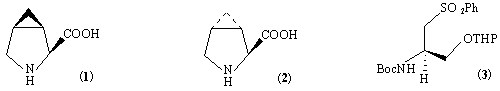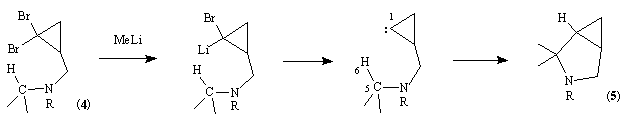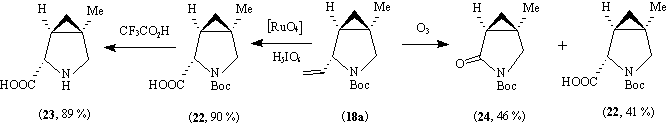Tetrahedron, Vol. 53, No 43, pp. 14773-14792, 1997
Copyright (c) 1997 Elsevier Science
Synthesis of (2S, 3R, 4S)-3,4-Methanoproline and Analogues
by Cyclopropylidene Insertion
Viacheslav V. Tverezovskyb, Mark S. Bairda* and Ivan G. Bolesovb
a Department of Chemistry, University of Wales, Bangor
b Department of Chemistry, State University of Moscow, Moscow
|
Abstract: Intramolecular insertion of single enantiomers of cyclopropylidenes into 5,6-related C-H bonds adjacent to nitrogen has been used to obtain enantiomerically pure methanoproline and a number of analogues with a high degree of one- or two-fold asymmetric induction.
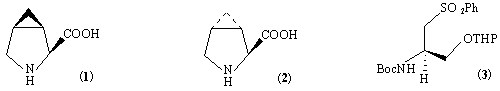
|
Although methanoproline (1) was isolated some years ago from the American horse chesnut, Aesculus parviflora,1 it is still attracting considerable attention.2,3 It was shown to be a potent inhibitor of proline metabolism;4 as such it was targeted as a potential chemical control agent in the production of hybrid wheats.5 In addition, methanoproline inhibits the proline transport system of Escherichia coli,6 while amide derivatives of methanoproline are inhibitors of angiotensin converting enzyme.7 Although a number of routes to racemic methanoproline have been reported,8 only one synthesis of enantiomerically pure (1)9 and one synthesis of the N-Boc derivative have appeared.10 The first involves the cyclopropanation of dehydroproline in a reaction which is not very diastereoselective and from which the major product is the trans-isomer (2).
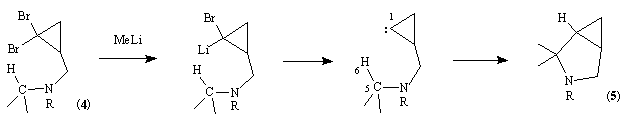
The second involves the coupling of two chiral starting materials, (3) and (2R)-glycidyl triflate, followed by cyclisation and functional group interconvertion to give N-Boc protected (1) in seven steps with overall yield of less then 25 %. We reported some years ago that 2-dialkylaminomethyl-1,1-dibromocyclopropanes such as (4) react with methyllithium to give 3-azabicyclo[3.1.0]hexanes (5) by a formal insertion of the derived cyclopropylidene or a related carbenoid into the CH-bond adjacent to nitrogen and 5,6-related to the carbene centre:11
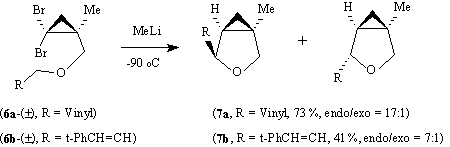
It has also been shown that a similar insertion of cyclopropylidenes derived from the ethers (6a) and (6b) by reaction with methyllithium leads to the 3-oxabicyclo[3.1.0]hexanes (7a) and (7b) with a moderate to high diastereoselectivity for the endo-R isomer.12
We now report the application of the insertion reaction to the synthesis of methanoproline and of a number of related compounds each as a single enantiomer. The method has the advantage that, although involving a number of steps, the starting materials are cheap and readily available on a large scale and the reactions involved are generally extremely high yielding; moreover it may be varied to produce in principle any required stereoisomer and a range of analogues.

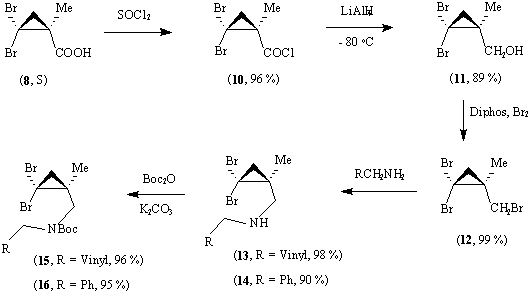
Reaction of methyl methacrylate with bromoform and base leads very readily to 2,2-dibromo-1-methylcyclopropane carboxylic acid;13 this is readily resolved using dehydroabietylamine in methanol to give (8, R) and its enantiomer, and the absolute stereochemistry of the amide of (8, R) has been established by X-ray crystallography.14 The corresponding non-methylated acid (9, R) and its enantiomer were readily obtained on a multi-gram scale by oxidation of 1,1-dibromo-2-vinylcyclopropane with potassium permanganate to give 2,2-dibromocyclopropanecarboxylic acid followed by resolution of this with dehydroabietylamine.14
The acid (8, S) (>99% e.e.) was converted into the corresponding acid chloride by reaction with thionyl chloride, and this was reduced to the alcohol (11) by reaction with lithium aluminium hydride. This route was chosen in order to avoid competing reduction of the dibromocyclopropane to the corresponding isomeric mono-bromides. It was necessary to add a solution of the acid chloride in ether to lithium aluminium hydride in ether at - 80 o
C, as addition of the hydride to the acid chloride led to up to 30 % of an ester derived by reaction of the product alcohol with the acid chloride. At higher temperatures monobromides were formed in addition to dibromi-des. The alcohol (11) was converted into the corresponding bromide (12) by reaction with 1,2-bis(diphenyl-phosphino)ethane and bromine, and reaction of this with either allylamine or benzylamine gave the amines (13) and (14). *
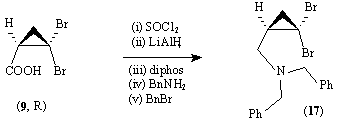
By a similar sequence of reactions, the acid (9, R) (93 % e.e.) was converted into the dibenzylamine (17) in an overall yield of 86 %. Attempts to prepare (17) from 1,1-dibromo-2-bromomethylcyclopropane derived from (9, R) directly by reaction with dibenzylamine led to only 80 % yield of (17) after 4 days at 50 o
C.
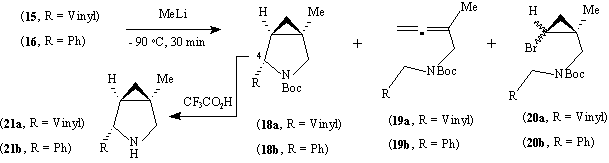
The amines (13) and (14) were protected as their Boc derivatives (15) and (16); reaction of these with methyl lithium led to a mixture of products, the major component of which was the azabicyclo[3.1.0]hexane (18a) or (18b) respectively. The yields of each of the products were highly dependent on the reaction conditions (Table 1). The stereochemistry of the allyl and phenyl groups in (18a) or (18b) was assigned on the basis of the fact that the signal for H-4 in the proton NMR spectrum in each case appeared as a singlet rather than the doublet expected for the epimer.12,15 Moreover, in each case these compounds showed the signals for two rotamers about the amide bond by 1H and 13C NMR; deprotection gave the amines (21a) and (21b) which showed only the expected number of signals in 1H and 13C NMR.
Table 1. Reactions of (15) and (16) with methyl lithium.
|
Starting
material |
Temperature during reaction, o
C a |
Compounds determined by NMR in reaction mixture, yields, % b |
| |
|
18 |
19 |
20 c |
unknown substancee |
starting material |
|
15 |
20 |
40 |
45 |
9 |
2 |
- |
| |
-30 |
50 |
19 |
17 |
7 |
3 |
| |
-60 |
61 |
8 |
16 |
2 |
1 |
| |
-90 |
49 |
8 |
29 |
8 |
1 |
|
16 |
20 |
40d |
27 |
7 |
1 |
11 |
| |
-90 |
25d |
8 |
17 |
3 |
24 |
a) Addition time was 5 min; stirring time was 30 min; b) these data were confirmed by glc; c) stereochemistry of monobromides was not determined; d) isolated yield; e) this might be the diastereomer of monobromide c.
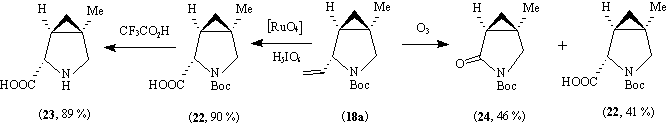
Although the purified yield of (18a) obtained in this way was only 47 %, it could readily be converted into the methanoproline analoge (23) by oxidation with ruthenium tetroxide, generated in situ from RuCl3, followed by deprotection. Attempted ozonolysis also led to (22) but in this case a second product was the lactam (24).
The amine (14) was benzylated by reaction with benzyl bromide to give (25). In contrast to (16), the diben-zylamine (25) reacted with methyllithium to give the endo-phenyl isomer (26) as the major product (Table 2):
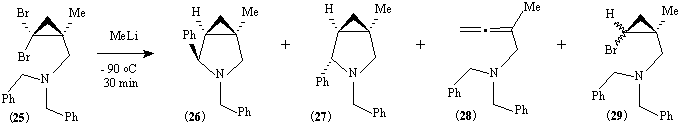
Table 2. Reaction of compound (25) with methyl lithium.
|
Temperature during
reaction, oC a |
Compounds determined by NMR in reaction mixture, yields, % b |
| |
26 |
27 |
28 |
29 c,f |
|
20 |
56 |
5 d |
33 |
- |
|
-90 |
90 |
1.8 d |
2 |
6 |
a, b, c, d see Table 1; f) strucure assigned on the basis of cyclopropane signals in the 1H NMR of the mixture of (26), (28) and (29).
The amine (26) could be separated from its bicyclic isomer (27) by chromatography (96%, >90 % pure). It could be obtained pure by removal of the impurites (28) and (29) by selective debenzylation with hydrogen and a catalyst (5 % Pd/C, 0.01 mol.eq., 1 h) followed by flash chromatography.
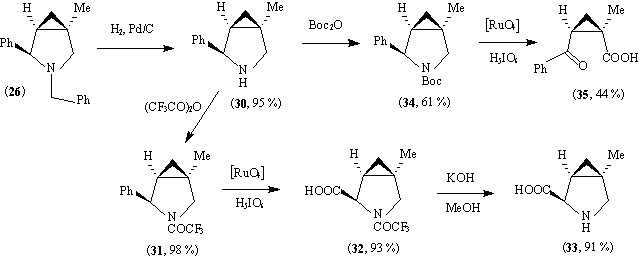
The crude amine (26) was debenzylated to (30) under more vigorous conditions (0.05 eq. of the same catalyst, 15 h). Protection by trifluoroacetylation, then oxidation of the phenyl group by reaction with ruthenium tetroxide at 80 o
C for 3 h gave (32), and then deprotection gave the aminoacid (33), a second methylated analogue of methanoproline. It is interesting to note the remarkable stability of CH and CH2 groups near nitrogen to oxidation under these conditions. It is well known that ethers RCH2OMe (R=Ph or Alk) can readily be oxidised to esters with ruthenium tetroxide even at room temperature.16 Attempts to oxidise (31) with ruthenium tetroxide at 20 o
C even using more catalyst and for 1 day were unsuccessful. In the case of the Boc - protected amine (34), ruthenium tetroxide oxidation at room temperature led to ring opening, to give (35).
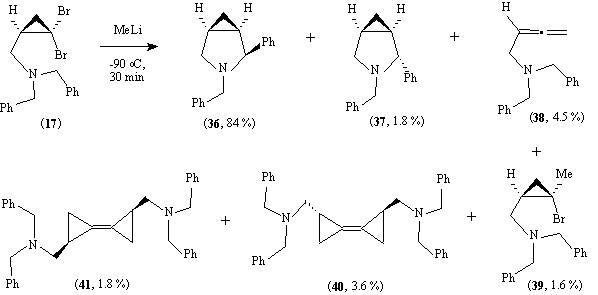
Reaction of (17) with methyl lithium gave one major product (36) together with a number of minor products with structures typical of those usually obtained in similar reactions of non-functionalised dihalocyclopropanes.17
The stereochemistry of the bicyclopropilydenes (40) and (41) is not certain, while that of (39) is assigned on the basis of NOE studies. The use of modified reaction conditions to increase the yields of novel optically active bicyclopropylidenes is being examined.
The reason for the reversal in the stereochemistry of the major azabicycles (26) and (36) compared to that of (18a) and (18b) is not yet clear, but may reflect the increased steric demand of the Boc group compared to benzyl, or more probably the steric and electronic effect of the N-CO bond of the intermediates leading to (18a) and (18b) compared to the N-CH2 bond of those leading to (26) and (36). Indeed, the NMR spectra of the products (18a) and (18b) are characterised by the presence of two rotamers about this bond.
Debenzylation of (36) (93 % e.e.) followed by protection as a trifluoroacetate gave crystalline (42) (81 % yield, 100% e.e. by chiral g.l.c., with baseline resolution, see acknowledgement). Oxidation as before and deprotec-tion gave the final aminoacid (1) with an NMR spectrum which was identical to that in the literature.9 The [ ]D20 (- 141.8o
) was also in good agreement with the literature value ([
]D20 (- 141.8o
) was also in good agreement with the literature value ([ ]D20 - 144o
).9 The overall yield of (1) from (9, R) was 41 %.
]D20 - 144o
).9 The overall yield of (1) from (9, R) was 41 %.
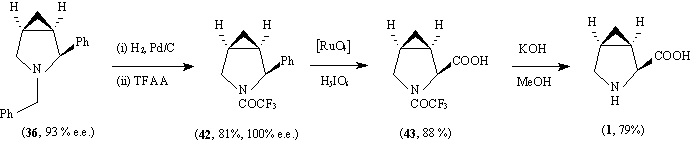
The reactions described above indicate that the insertion of cyclopropylidenes into C-H bonds can occur in a highly stereocontrolled manner and may offer a valuable route to a number of optically active mono- and polycyclic systems.
Acknowledgement: This work was carried out with the support of the INTAS programme.
We thank Dr. Nikolay S. Ikonnikov, Nesmeyanov Institute of Organoelement Compounds, 117813,
28 Vavilov, Moscow, for kindly supplying a chiral g.l.c. column which provided excellent resolution.
Experimental Section
Reagents were obtained from commercial suppliers and were used without further purification unless stated. Dichloromethane was distilled over calcium hydride. Diethyl ether and tetrahydrofuran were distilled over sodium wire. Petroleum was either of boiling point 40 - 60 o
C or 60 - 80 o
C and was distilled. Reactions requiring anhydrous conditions were performed using oven dried glassware (250 o
C) that was cooled under either dry nitrogen or argon and the experiments were conducted under a positive atmosphere of one of these gases. Organic solutions were dried over anhydrous magnesium sulphate, and, unless stated, were evaporated at 14 mmHg. Yields quoted are for the purified compounds unless stated.
All new compounds were homogeneous by tlc or by glc. Glc was conducted using a Perkin-Elmer Model F17 F.I.D. on a capillary column (30 m x 0.32 mm id Phase, DB5 split ratio of 50:1) using nitrogen as carrier gas. Chiral glc was conducted using a 2,6-diamil-3-trifluoroacetyl-a-cyclodextrin fused silica column(40 m x 0.23 mm ID, film 0.12 mm), using helium as a carrier gas at 2 bar pressure (see acknowledgement). Tlc was performed using Aldrich silica gel 60 plates (F254). Compounds were visualised either by examination under an ultraviolet source or by exposure to iodine vapour. Column chromatography was conducted with Merck 7736 silica gel under medium pressure.
Melting points are uncorrected. Infrared spectra were obtained on a Perkin-Elmer 1600 FTIR spectrometer as liquid filmsunless otherwise stated. Low resolution mass spectra were obtained using a Finnigan Mat 1020 spectrometer. Mass measurements refer to 79Br and 35Cl isotopes unless stated and were obtained from the Swansea Mass Spectrometry Service. Microanalyses were performed with a Carlo-Erba Model 1106 CHN analyser. NMR spectra were recorded in CDCl3 unless otherwise stated on a Bruker AC250 at 250 MHz for protons and 62.9 MHz for carbon and in the latter case were broad-band decoupled. In most cases DEPT spectra were also run and the signs of signals (+ for CH, CH3; - for CH2) are indicated on the data for the broad-band decoupled spectrum. Those signals with no sign in such a spectrum are quaternary.
(S)-(+)-2,2-Dibromo-1-methylcyclopropylcarbonyl chloride.
Thionyl chloride (25 ml, 0.34 mole) was added to (S)-(-)-2,2-dibromo-1-methylcyclopropane carboxylic acid (4, S), [ ]D20 -55.1o
(c 1.015, CHCl3), >99% e.e.14 (26.4 g, 102.4 mmole) and the mixture was refluxed for 2 h. The excess of thionyl chloride was removed by distillation and the residue was distilled at 39 - 40 o
C (0.8 mmHg) to give (S)-(+)-2,2-dibromo-1-methylcyclopropylcarbonyl chloride (10) (27.2 g, 98.4 mmole, 96 %), [
]D20 -55.1o
(c 1.015, CHCl3), >99% e.e.14 (26.4 g, 102.4 mmole) and the mixture was refluxed for 2 h. The excess of thionyl chloride was removed by distillation and the residue was distilled at 39 - 40 o
C (0.8 mmHg) to give (S)-(+)-2,2-dibromo-1-methylcyclopropylcarbonyl chloride (10) (27.2 g, 98.4 mmole, 96 %), [ ]D20 +0.3o
(c 1.104, CHCl3), m.p. -15 o
C, which showed
]D20 +0.3o
(c 1.104, CHCl3), m.p. -15 o
C, which showed  H: 1.77 (3H, s), 1.78 (1H, d, J 8.0 Hz), 2.52 (1H, d, J 8.0 Hz);
H: 1.77 (3H, s), 1.78 (1H, d, J 8.0 Hz), 2.52 (1H, d, J 8.0 Hz);  C: 21.76+, 27.61, 34.37-, 42.97, 156.35; n
max: 1783 s, 1451 m, 1421 m, 1384 m, 1297 m, 1240 m, 1088 m, 1056 m, 1029 m, 951 s, 931 m, 790 s, 721 m, 655 m, 626 m cm-1.
C: 21.76+, 27.61, 34.37-, 42.97, 156.35; n
max: 1783 s, 1451 m, 1421 m, 1384 m, 1297 m, 1240 m, 1088 m, 1056 m, 1029 m, 951 s, 931 m, 790 s, 721 m, 655 m, 626 m cm-1.
(S)-(+)-2,2-Dibromo-1-hydroxymethyl-1-methylcyclopropane.
A solution of (S)-(+)-2,2-dibromo-1-methylcyclopropanecarbonyl chloride (24.7 g, 89.4 mmole) in dry ether (20 ml) was added with stirring to lithium aluminium hydride (3.66 g, 89.4 mmole) in dry ether (400 ml) at -85 o
C under nitrogen over 20 min. After 10 min at -85 o
C, ethyl acetate (40 ml) was slowly added and the mixture was warmed to 0 o
C and sat.aq. ammonium chloride (100 ml) was added. The aqueous layer was extracted with ether (100 ml). The combined organic layers were dried and solvent was removed to give crude product (21.7 g), which was sublimed at 5 mmHg and 70 o
C to yield (S)-(+)-2,2-dibromo-1-hydroxymethyl-1-methylcyclopropane (11), (19.5 g, 80.0 mmole, 89 %), (Found: C 24.41, H 3.50. Calculated for C4H8Br2O: C 24.62, H 3.31), m.p. 53 - 54 o
C, [ ]D20 +18.8o
(c 1.050, CHCl3) (for racemate see ref. 18) which showed
]D20 +18.8o
(c 1.050, CHCl3) (for racemate see ref. 18) which showed  H: 1.47 (1H, d, J 7.6), 1.51 (3H, s), 1.62 (1H, d, J 7.6), 1.89 (1H, dd, J 8.3, 3.9), 3.70 (1H, dd, J 11.9, 3.9), 3.86 (1H, dd, J 11.9, 8.3);
H: 1.47 (1H, d, J 7.6), 1.51 (3H, s), 1.62 (1H, d, J 7.6), 1.89 (1H, dd, J 8.3, 3.9), 3.70 (1H, dd, J 11.9, 3.9), 3.86 (1H, dd, J 11.9, 8.3);  C: 20.52+, 31.39, 32.55-, 35.75, 69.84-; n
max: 3258 broad s, 2931 m, 1453 m, 1050 s, 1037 s, 1023 s, 787 s, 763 s, 691 s cm-1.
C: 20.52+, 31.39, 32.55-, 35.75, 69.84-; n
max: 3258 broad s, 2931 m, 1453 m, 1050 s, 1037 s, 1023 s, 787 s, 763 s, 691 s cm-1.
(S)-(+)-2,2-Dibromo-1-bromomethyl-1-methylcyclopropane.
A solution of bromine (0.55 ml, 16.5 mmole) in dichloromethane (2 ml) was slowly added to diphos (3.44 g, 8.63 mmole) in dichloromethane (25 ml) at 0 o
C. (S)-(+)-2,2-Dibromo-1-hydroxymethyl-1-methylcyclopropane (3.66 g, 15.0 mmole) in dichloromethane (15 ml) was then added at 0 o
C and the mixture was stirred for 3 h. Ether
(50 ml) was added, the solution was filtered and solvent was removed to give crude product which was purified by short column chromatography over silica eluting with petrol and ether (4:1) to yield (S)-(+)-2,2-dibromo-1-bromomethyl-1-methylcyclopropane (12), (4.54 g, 14.8 mmole, 99 %), [ ]D20 +26.4o
(c 0.884, CHCl3) (for racemate see ref. 20) which showed
]D20 +26.4o
(c 0.884, CHCl3) (for racemate see ref. 20) which showed  H: 1.57 (3H, s), 1.66 (1H, d, J 7.8 Hz), 1.70 (1H, d, J 7.8 Hz), 3.55 (1H, d, J 10.5 Hz), 3.73 (1H, d, J 10.5 Hz);
H: 1.57 (3H, s), 1.66 (1H, d, J 7.8 Hz), 1.70 (1H, d, J 7.8 Hz), 3.55 (1H, d, J 10.5 Hz), 3.73 (1H, d, J 10.5 Hz);  C: 22.21+, 30.31, 35.68-, 36.68, 41.97-; n
max: 2994 s, 2965 s, 2929 s, 2868 m, 1451 s, 1427 s, 1381 s, 1320 m, 1270 m, 1227 s, 1150 m, 1078 s, 1048 s, 1026 s, 959 s, 900 m, 866 m, 839 m, 703 s, 642 s cm-1.
C: 22.21+, 30.31, 35.68-, 36.68, 41.97-; n
max: 2994 s, 2965 s, 2929 s, 2868 m, 1451 s, 1427 s, 1381 s, 1320 m, 1270 m, 1227 s, 1150 m, 1078 s, 1048 s, 1026 s, 959 s, 900 m, 866 m, 839 m, 703 s, 642 s cm-1.
(S)-(-)-N-Allyl-2,2-dibromo-1-methylcyclopropylmethylamine.
(S)-(+)-2,2-Dibromo-1-bromomethyl-1-methylcyclopropane (1.53 g, 5.0 mmole) was added to allylamine (3.8 ml, 50 mmole) in benzene (5 ml) and refluxed for 15 h, then extracted with 5 % aq. sodium hydroxide (5 ml, 6 mmole) and washed with brine (5 ml). The organic layer was dried and the solvent was removed to give (S)-(-)-N-allyl-2,2-dibromo-1-methylcyclopropylmethylamine (13), (1.38 g, 4.88 mmole, 98 %), [ ]D20-15.4o
(c 0.968, CHCl3) which showed
]D20-15.4o
(c 0.968, CHCl3) which showed  H: 1.40 (1H, s), 1.43 (1H, d, J 7.4 Hz), 1.48 (3H, s), 1.53 (1H, d, J 7.4 Hz), 2.75 (1H, d, J 12.4 Hz), 2.86 (1H, d, J 12.4 Hz), 3.28 (1H, ddd, J 6.0, 1.6, 1.0 Hz), 5.11 (1H, ddt, J 10.2, 1.6, 1.0 Hz), 5.20 (1H, ddt, J 17.1, 1.6, 1.6 Hz), 5.91 (1H, ddt, J 17.1, 10.2, 6.0 Hz);
H: 1.40 (1H, s), 1.43 (1H, d, J 7.4 Hz), 1.48 (3H, s), 1.53 (1H, d, J 7.4 Hz), 2.75 (1H, d, J 12.4 Hz), 2.86 (1H, d, J 12.4 Hz), 3.28 (1H, ddd, J 6.0, 1.6, 1.0 Hz), 5.11 (1H, ddt, J 10.2, 1.6, 1.0 Hz), 5.20 (1H, ddt, J 17.1, 1.6, 1.6 Hz), 5.91 (1H, ddt, J 17.1, 10.2, 6.0 Hz);  C: 21.78+, 29.90, 33.55-, 37.31, 52.39-, 57.13-, 116.04-, 136.77+; n
max: 3076 m, 2967 s, 2927 s, 2849 s, 2820 s, 1643 m, 1455 s, 1428 m, 1381 m, 1148 m, 1114 s, 1071 s, 1040 s, 1020 s, 993 s, 919 s, 759 m, 690 s cm-1.
C: 21.78+, 29.90, 33.55-, 37.31, 52.39-, 57.13-, 116.04-, 136.77+; n
max: 3076 m, 2967 s, 2927 s, 2849 s, 2820 s, 1643 m, 1455 s, 1428 m, 1381 m, 1148 m, 1114 s, 1071 s, 1040 s, 1020 s, 993 s, 919 s, 759 m, 690 s cm-1.
(S)-(-)-N-Benzyl-2,2-dibromo-1-methylcyclopropylmethylamine.
A solution of (S)-(+)-2,2-dibromo-1-bromomethyl-1-methylcyclopropane (1.84 g, 6.0 mmole) in DMSO (5 ml) was added to benzylamine (1.93 g, 18.0 mmole) in DMSO (10 ml) and stirred for 20 h. It was diluted with water (60 ml) and extracted with ether (3 x 25 ml) and the organic layers were combined and dried. After removing the solvent, the crude product was purified by column chromatography over silica to give starting material (0.14 g, 0.5 mmole) and (S)-(-)-N-benzyl-2,2-dibromo-1-methylcyclopropylmethylamine (14), (1.80 g, 5.41 mmole, 98 %, based on starting material consumed), [ ]D20-8.0o
(c 0.934, CHCl3) which showed
]D20-8.0o
(c 0.934, CHCl3) which showed  H: 1.41 (1H, d, J 7.5 Hz), 1.48 (3H, s), 1.49 (1H, d, J 7.5 Hz), 1.58 (1H, s), 2.72 (1H, d, J 12.4 Hz), 2.86 (1H, d, J 12.4 Hz), 3.81 (2H, s), 7.20-7.37 (5H, m);
H: 1.41 (1H, d, J 7.5 Hz), 1.48 (3H, s), 1.49 (1H, d, J 7.5 Hz), 1.58 (1H, s), 2.72 (1H, d, J 12.4 Hz), 2.86 (1H, d, J 12.4 Hz), 3.81 (2H, s), 7.20-7.37 (5H, m);  C: 21.87+, 30.05, 33.53-, 37.45, 53.93-, 57.31-, 127.03+, 128.11+, 128.41+, 140.29; n
max: 3026 m, 2989 m, 2927 m, 2890 m, 2842 m, 1494 m, 1453 s, 1113 m, 1071 m, 1042 m, 1027 m, 739 s, 698 s cm-1.
C: 21.87+, 30.05, 33.53-, 37.45, 53.93-, 57.31-, 127.03+, 128.11+, 128.41+, 140.29; n
max: 3026 m, 2989 m, 2927 m, 2890 m, 2842 m, 1494 m, 1453 s, 1113 m, 1071 m, 1042 m, 1027 m, 739 s, 698 s cm-1.
(S)-(+)-N-t-Butoxycarbonyl-N-allyl-2,2-dibromo-1-methylcyclopropylmethylamine.
Potassium carbonate (0.74 g, 7.0 mmole) was added to (S)-(-)-N-allyl-2,2-dibromo-1-methylcyclo-propylmethylamine (0.99 g, 3.50 mmole) and t-butoxycarbonylanhydride (0.96 g, 4.38 mmole) in THF (10 ml). The mixture was vigorously stirred for 2 h, then filtered and THF was evaporated. A solution of sodium hydroxide (0.12 g, 3 mmole) in ethanol (10 ml) was added to the residue and the mixture was stirred for 1 h. The solvent was removed and the crude product was purified by short column chromatography over silica eluting with petrol and ether (3:1) to yield (S)-(+)-N-t-butoxycarbonyl-N-allyl-2,2-dibromo-1-methylcyclopropylmethylamine (15), (1.29 g, 3.37 mmole, 96 %), [ ]D20 +22.6o
(c 1.116, CHCl3) which showed
]D20 +22.6o
(c 1.116, CHCl3) which showed  H: 1.36 (3H, s), 1.49 (10H, s), 1.54 (1H, d, J 8.5 Hz), 3.30-3.90 (3H, br.m), 3.99 (1H, dd, J 10.2, 5.5 Hz), 5.11-5.19 (2H, m), 5.82 (1H, ddt, J 17.0, 10.4, 5.5 Hz);
H: 1.36 (3H, s), 1.49 (10H, s), 1.54 (1H, d, J 8.5 Hz), 3.30-3.90 (3H, br.m), 3.99 (1H, dd, J 10.2, 5.5 Hz), 5.11-5.19 (2H, m), 5.82 (1H, ddt, J 17.0, 10.4, 5.5 Hz);  C: 21.00+, 28.38+, (33.24, 33.80)-, 37.27, 48.98-, (51.90, 52.37)-, 79.86, 116.25-, 133.63+; n
max: 2975 s, 2931 m, 1698 s, 1455 s, 1408 s, 1365 s, 1319 m, 1245 s, 1172 s, 1148 s, 955 m, 923 m, 873 m, 774 m, 694 m cm-1.
C: 21.00+, 28.38+, (33.24, 33.80)-, 37.27, 48.98-, (51.90, 52.37)-, 79.86, 116.25-, 133.63+; n
max: 2975 s, 2931 m, 1698 s, 1455 s, 1408 s, 1365 s, 1319 m, 1245 s, 1172 s, 1148 s, 955 m, 923 m, 873 m, 774 m, 694 m cm-1.
(S)-(+)-N-t-Butoxycarbonyl-N-benzyl-2,2-dibromo-1-methylcyclopropylmethylamine.
Potassium bicarbonate (0.34 g, 4.0 mmole) was added to (S)-(-)-N-benzyl-2,2-dibromo-1-methyl-cyclopropylmethylamine (0.27 g, 0.81 mmole) and t-butoxycarbonylanhydride (0.22 g, 1.01 mmole) in THF (5 ml). The mixture was vigorously stirred for 18 h, then filtered and THF was evaporated. Work up as above and short column chromatography over silica eluting with petrol and ether (2:1) gave (S)-(+)-N-t-butoxycarbonyl-N-benzyl-2,2-dibromo-1-methylcyclopropylmethylamine (16), (0.33 g, 0.77 mmole, 95 %), [ ]D20 +16.0o
(c 1.373, CHCl3) which showed
]D20 +16.0o
(c 1.373, CHCl3) which showed  H: 1.34 - 1.50 (14H, m), 3.20 - 3.70 (2H, br.m), 4.43 (1H, br.d, J 15.8 Hz), 4.57 (1H, d, J 15.8 Hz), 7.18 - 7.58 (5H, m);
H: 1.34 - 1.50 (14H, m), 3.20 - 3.70 (2H, br.m), 4.43 (1H, br.d, J 15.8 Hz), 4.57 (1H, d, J 15.8 Hz), 7.18 - 7.58 (5H, m);  C: 21.07+, 28.40+, (33.46, 33.90)-, 36.02, (49.85, 49.98)-, 52.38-, 80.29, 127.11+, 127.22+, 128.48+, 138.37, (156.06, 156.36); n
max: 3064 m, 3030 m, 2969 s, 2929 s, 1700 br.s, 1496 m, 1454 s, 1416 s, 1366 s, 1309 s, 1250 s, 1180 s, 1122 s, 1082 m, 1066 m, 1028 m, 960 m, 902 m, 879 m, 768 m, 698 s cm-1.
C: 21.07+, 28.40+, (33.46, 33.90)-, 36.02, (49.85, 49.98)-, 52.38-, 80.29, 127.11+, 127.22+, 128.48+, 138.37, (156.06, 156.36); n
max: 3064 m, 3030 m, 2969 s, 2929 s, 1700 br.s, 1496 m, 1454 s, 1416 s, 1366 s, 1309 s, 1250 s, 1180 s, 1122 s, 1082 m, 1066 m, 1028 m, 960 m, 902 m, 879 m, 768 m, 698 s cm-1.
(R)-(+)-2,2-Dibromocyclopropylcarbonyl chloride.
Thionyl chloride (12 ml, 164 mmole) was added to (R)-(+)-2,2-dibromocyclopropanecarboxylic acid (5, R), ([ ]D20 +129.4o
(c 1.036, CHCl3), 92.5% e.e.14), (10.00 g, 41.0 mmole) and the mixture was refluxed for 2 h. The excess of thionyl chloride was removed by distillation to give (R)-(+)-2,2-dibromocyclopropylcarbonyl chloride (10.72 g, 40.9 mmole, 100 %), [
]D20 +129.4o
(c 1.036, CHCl3), 92.5% e.e.14), (10.00 g, 41.0 mmole) and the mixture was refluxed for 2 h. The excess of thionyl chloride was removed by distillation to give (R)-(+)-2,2-dibromocyclopropylcarbonyl chloride (10.72 g, 40.9 mmole, 100 %), [ ]D20 +109.7o
(c 1.068, CHCl3) which show
]D20 +109.7o
(c 1.068, CHCl3) which show  H: 2.18 (1H, dd, J 9.2, 7.9 Hz), 2.27 (1H, dd, J 7.9, 7.6 Hz), 3.12 (1H, dd, J 9.2, 7.6 Hz);
H: 2.18 (1H, dd, J 9.2, 7.9 Hz), 2.27 (1H, dd, J 7.9, 7.6 Hz), 3.12 (1H, dd, J 9.2, 7.6 Hz);  C: 19.34, 31.18-, 42.63+, 168.06; n
max: 1779 br. s, 1417 m, 1347 s, 1104 m, 1069 s, 1001 s, 940 m, 836 s, 741 m, 689 s, cm-1.
C: 19.34, 31.18-, 42.63+, 168.06; n
max: 1779 br. s, 1417 m, 1347 s, 1104 m, 1069 s, 1001 s, 940 m, 836 s, 741 m, 689 s, cm-1.
(R)-(+)-2,2-Dibromo-1-hydroxymethylcyclopropane.
A solution of (R)-(+)-2,2-dibromocyclopropylcarbonyl chloride (10.68 g, 40.7 mmole) in dry ether (20 ml) was added to a stirred solution of lithium aluminium hydride (1.64 g, 40 mmole) in dry ether (150 ml) at -85 o
C under nitrogen over 30 min. After 10 min at -85 o
C, work up as for (6) and removal of the solvent gave (R)-(+)-2,2-dibromo-1-hydroxymethylcyclopropane (9.02 g, 39.2 mmole, 96 %) as colourless oil, [ ]D20 +2.4o
(c 1.234, CHCl3) (for racemate see ref. 21) which showed
]D20 +2.4o
(c 1.234, CHCl3) (for racemate see ref. 21) which showed  H: 1.43 (1H, dd, J 7.5, 7.3 Hz), 1.82 (1H, dd, J 10.4, 7.3 Hz), 1.97 (1H, dddd, J 10.4, 8.5, 7.5, 5.1 Hz), 2.33 (1H, s), 3.62 (1H, dd, J 12.2, 8.5 Hz), 3.91 (1H, dd, J 12.2, 5.1 Hz);
H: 1.43 (1H, dd, J 7.5, 7.3 Hz), 1.82 (1H, dd, J 10.4, 7.3 Hz), 1.97 (1H, dddd, J 10.4, 8.5, 7.5, 5.1 Hz), 2.33 (1H, s), 3.62 (1H, dd, J 12.2, 8.5 Hz), 3.91 (1H, dd, J 12.2, 5.1 Hz);  C: 26.01, 26.45-, 32.33+, 64.84-; nmax : 3346 br. s, 2934 m, 2879 m, 1462 m, 1432 m, 1390 m, 1240 m, 1103 s, 1042 s, 682 s cm-1.
C: 26.01, 26.45-, 32.33+, 64.84-; nmax : 3346 br. s, 2934 m, 2879 m, 1462 m, 1432 m, 1390 m, 1240 m, 1103 s, 1042 s, 682 s cm-1.
(R)-(-)-2,2-Dibromo-1-bromomethylcyclopropane.
A solution of bromine (2.21 ml, 42.9 mmole) in dichloromethane (5 ml) was slowly added to a solution of diphos (8.93 g, 22.4 mmole) in dichloromethane (150 ml) at 0 o
C. (R)-(+)-2,2-Dibromo-1-hydroxymethyl-cyclopropane (8.99 g, 39.1 mmole) in dichloromethane (30 ml) was then added at 0 o
C and the reaction mixture was stirred for 3 h. The solution was evaporated to a volume of 100 ml and ether (100 ml) was added, and the solution was separated from a solid. The solid was dissolved in dichloromethane (50 ml) and ether (100 ml) was added. The solution was separated from precipitate and the last procedure was repeated 4 times, until there was no product in the organic layer by glc. All solutions were combined and the solvent was removed to give a crude product which was purified by short column chromatography over silica eluting with petrol and ether (4:1) to yield (R)-(-)-2,2-dibromo-1-bromomethylcyclopropane (11.08 g, 37.8 mmole, 97 %), [ ]D20 -15.9o
(c 0.962, CHCl3) (for racemate see refs. 20, 22) which showed
]D20 -15.9o
(c 0.962, CHCl3) (for racemate see refs. 20, 22) which showed  H: 1.46 (1H, dd, J 7.7, 7.4 Hz), 1.95 (1H, dd, J 10.1, 7.4 Hz), 2.10 (1H, ddt, J 10.1, 7.7, 7.5 Hz), 3.48 (2H, d, J 7.5 Hz);
H: 1.46 (1H, dd, J 7.7, 7.4 Hz), 1.95 (1H, dd, J 10.1, 7.4 Hz), 2.10 (1H, ddt, J 10.1, 7.7, 7.5 Hz), 3.48 (2H, d, J 7.5 Hz);  C: 20.62, 23.58-, 25.61+, 26.84-; n
max: 1430 s, 1366 s, 1230 m, 1215 s, 1191 m, 1106 s, 1047 m, 1011 m, 937 m, 690 s, 621 m cm-1.
C: 20.62, 23.58-, 25.61+, 26.84-; n
max: 1430 s, 1366 s, 1230 m, 1215 s, 1191 m, 1106 s, 1047 m, 1011 m, 937 m, 690 s, 621 m cm-1.
(R)-(+)-N-Benzyl-2,2-dibromocyclopropylmethylamine.
A solution of (R)-(-)-2,2-dibromo-1-bromomethylcyclopropane (5.86 g, 20.0 mmole) in DMSO (20 ml) was added to benzylamine (6.43 g, 60.0 mmole) in DMSO (20 ml). The mixture was stirred for 4 h at 60 o
C, diluted with water (100 ml), and 5 % aq. of sodium hydroxide (20 ml, 25 mmole) was added and the mixture was extracted with ether (3 x 100 ml). The organic layers were combined and dried. After removing the solvent, the crude product was purified by chromatography over silica (petrol-ether, 1:1) to give (R)-(+)-N-benzyl-2,2-dibromo-cyclopropylmethylamine (6.01 g, 18.8 mmole, 94 %), (Found: M+, 318.9394. Calculated for C11H13N79Br81Br: 318.9394), [ ]D20 +1.5o
(c 1.348, CHCl3) which showed
]D20 +1.5o
(c 1.348, CHCl3) which showed  H: 1.34 (1H, dd, J 7.1, 6.8 Hz), 1.75 (1H, s), 1.78 (1H, dd, J 10.4, 6.8 Hz), 1.86 (1H, dddd, J 10.4, 8.0, 7.1, 5.2 Hz), 2.72 (1H, dd, J 12.8, 8.0 Hz), 2.89 (1H, dd, J 12.8, 5.2 Hz), 3.87 (1H, d, J 13.1 Hz), 3.89 (1H, d, J 13.1 Hz), 7.26 - 7.38 (5H,m) ;
H: 1.34 (1H, dd, J 7.1, 6.8 Hz), 1.75 (1H, s), 1.78 (1H, dd, J 10.4, 6.8 Hz), 1.86 (1H, dddd, J 10.4, 8.0, 7.1, 5.2 Hz), 2.72 (1H, dd, J 12.8, 8.0 Hz), 2.89 (1H, dd, J 12.8, 5.2 Hz), 3.87 (1H, d, J 13.1 Hz), 3.89 (1H, d, J 13.1 Hz), 7.26 - 7.38 (5H,m) ;  C: 15.34, 27.29-, 30.94+, 52.00-, 53.67-, 127.13+, 128.16+, 128.50+, 139.92; n
max: 3061 m, 3026 s, 2891 m, 2830 s, 1494 m, 1454 s, 1358 m, 1108 s, 1044 m, 1028 m, 734 s, 698 s, 679 s cm-1; (m\z, %): 321, 2; 320, 2; 319, 3; 318, 3; 317, 2 (M+); 316, 2; 255, 2; 146, 20; 100, 24; 91, 100.
C: 15.34, 27.29-, 30.94+, 52.00-, 53.67-, 127.13+, 128.16+, 128.50+, 139.92; n
max: 3061 m, 3026 s, 2891 m, 2830 s, 1494 m, 1454 s, 1358 m, 1108 s, 1044 m, 1028 m, 734 s, 698 s, 679 s cm-1; (m\z, %): 321, 2; 320, 2; 319, 3; 318, 3; 317, 2 (M+); 316, 2; 255, 2; 146, 20; 100, 24; 91, 100.
(R)-(+)-N,N-Dibenzyl-2,2-dibromocyclopropylmethylamine.
Sodium carbonate (3.71 g, 35.0 mmole) in water (20 ml) was added to (R)-(+)-N-benzyl-2,2-dibromocyclopropylmethylamine (5.42 g, 17.0 mmole) and benzylbromide (6.0 g, 35 mmole) in dichloromethane (20 ml) and the mixture was refluxed and stirred for 15 h. The water layer was extracted with dichloromethane (2 x 5 ml). The organic layers were combined, dried and the solvent was removed. Most of the benzylbromide was evaporated in vacuo (100 o
C, 1 mmHg). The residue was purified by chromatography over silica (petrol - ether, 2:98) to give (R)-(+)-N,N-dibenzyl-2,2-dibromocyclopropylmethylamine (17) (6.80 g, 16.6 mmole, 98 %) as colourless, very viscous oil (Found: M+, 408.9864. Calculated for C18H19N79Br81Br: 408.9864), [ ]D20 +17.0o
(c 1.024, CHCl3) which showed
]D20 +17.0o
(c 1.024, CHCl3) which showed  H: 1.23 (1H, dd, J 7.2, 5.8 Hz), 1.81 (1H, dd, J 10.6, 5.8 Hz), 1.83 (1H, dddd, J 10.6, 7.2, 6.4, 5.1 Hz), 2.66 (1H, dd, J 13.7, 5.1 Hz), 2.81 (1H, dd, J 13.7, 6.4 Hz), 3.65 (2H, d, J 13.6 Hz), 3.80 (2H, d, J 13.6 Hz), 7.25 - 7.45 (10H, m);
H: 1.23 (1H, dd, J 7.2, 5.8 Hz), 1.81 (1H, dd, J 10.6, 5.8 Hz), 1.83 (1H, dddd, J 10.6, 7.2, 6.4, 5.1 Hz), 2.66 (1H, dd, J 13.7, 5.1 Hz), 2.81 (1H, dd, J 13.7, 6.4 Hz), 3.65 (2H, d, J 13.6 Hz), 3.80 (2H, d, J 13.6 Hz), 7.25 - 7.45 (10H, m);  C: 27.60-, 28.43, 29.09+, 55.77-, 58.30-, 127.03+, 128.30+, 128.80+, 139.41; n
max: 3084 m, 3061 m, 3026 s, 2925 m, 2801 s, 1494 s, 1453 s, 1368 s, 1247 m, 1110 s, 1074 m, 1028 s, 973 m, 746 s, 698 s, 678 s, cm-1; (m\z, %): 411, 3; 410, 3; 409, 4; 408, 4; 407, 3 (M+); 406, 1; 210, 30; 149, 4; 92, 12; 91, 100.
C: 27.60-, 28.43, 29.09+, 55.77-, 58.30-, 127.03+, 128.30+, 128.80+, 139.41; n
max: 3084 m, 3061 m, 3026 s, 2925 m, 2801 s, 1494 s, 1453 s, 1368 s, 1247 m, 1110 s, 1074 m, 1028 s, 973 m, 746 s, 698 s, 678 s, cm-1; (m\z, %): 411, 3; 410, 3; 409, 4; 408, 4; 407, 3 (M+); 406, 1; 210, 30; 149, 4; 92, 12; 91, 100.
(1R,4S,5S)-N-t-Butoxycarbonyl-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane.
A 1.5M solution of methyllithium in ether (3.03 ml, 4.55 mmole) was added to (S)-(+)-N-t-butoxycarbonyl-N-allyl-2,2-dibromo-1-methylcyclopropylmethylamine (1.66 g, 4.33 mmole) in dry ether (50 ml) under nitrogen at - 60 o
C over 5 min. The solution was stirred for 30 min then sat. aq. ammonium chloride (20 ml) was added. The water layer was extracted with ether (50 ml). The combined organic layers were evaporated and purified by chromatography over silica (ether-petrol, 1:9) to give (1R, 4S, 5S)-N-t-butoxycarbonyl-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane (18a), (453 mg, 2.03 mmole, 47 %) as mixture of two rotamers in ratio 1.5:1, [ ]D20-38.2o
(c 0.976, CHCl3) which showed n
max: 2976 m, 1699 s, 1449 m, 1405 s, 1388 s, 1366 m, 1176 m, 1123 m, 883 m cm-1; (m/z, %): 213, 9; 212, 10 (M+); 198, 12; 197, 14; 169, 44; 168, 59; 167, 38; 142, 27; 141, 38; 140, 13; 108, 20; 96, 24; major rotamer,
]D20-38.2o
(c 0.976, CHCl3) which showed n
max: 2976 m, 1699 s, 1449 m, 1405 s, 1388 s, 1366 m, 1176 m, 1123 m, 883 m cm-1; (m/z, %): 213, 9; 212, 10 (M+); 198, 12; 197, 14; 169, 44; 168, 59; 167, 38; 142, 27; 141, 38; 140, 13; 108, 20; 96, 24; major rotamer,  H: 0.39 (1H, m), 0.54 (1H, m), 1.07 (1H, m), 1.25 (3H, s), 1.41 (9H, s), 3.16 (1H, d, J 10.6 Hz), 3.61 (1H, d, J 10.6 Hz), 4.17 (1H, d, J 6.5 Hz), 5.03-5.18 (2H, m), 5.69-5.88 (1H, m);
H: 0.39 (1H, m), 0.54 (1H, m), 1.07 (1H, m), 1.25 (3H, s), 1.41 (9H, s), 3.16 (1H, d, J 10.6 Hz), 3.61 (1H, d, J 10.6 Hz), 4.17 (1H, d, J 6.5 Hz), 5.03-5.18 (2H, m), 5.69-5.88 (1H, m);  C: 15.39-, 18.64+, 22.07, 27.51+, 28.49+, 51.78-, 62.20+, 79.21, 114.22-, 138.00+, 154.73; minor rotamer,
C: 15.39-, 18.64+, 22.07, 27.51+, 28.49+, 51.78-, 62.20+, 79.21, 114.22-, 138.00+, 154.73; minor rotamer,  H: 0.39 (1H, m), 0.54 (1H, m), 1.07 (1H, m), 1.23 (3H, s), 1.43 (9H, s), 3.17 (1H, d, J 10.6 Hz), 3.55 (1H, d, J 10.6 Hz), 4.32 (1H, d, J 5.9 Hz), 5.03-5.18 (2H, m), 5.69-5.88 (1H, m);
H: 0.39 (1H, m), 0.54 (1H, m), 1.07 (1H, m), 1.23 (3H, s), 1.43 (9H, s), 3.17 (1H, d, J 10.6 Hz), 3.55 (1H, d, J 10.6 Hz), 4.32 (1H, d, J 5.9 Hz), 5.03-5.18 (2H, m), 5.69-5.88 (1H, m);  C: 15.31-, 18.55+, 22.78, 26.91+, 28.43+, 52.41-, 61.51+, 79.21, 114.31-, 137.53+, 154.51.
C: 15.31-, 18.55+, 22.78, 26.91+, 28.43+, 52.41-, 61.51+, 79.21, 114.31-, 137.53+, 154.51.
Also were isolated N-allyl-N-t-butoxycarbonyl-N-2-methylbuta-2,3-dienylamine (19a) (58 mg, 0.26 mmole, 6 %) (which showed  H: 1.48 s (9H), 1.68 (3H, t, J 3.1), 3.72 (4H, br. s), 4.65 (2H, m), 5.12 (2H, m), 5.75 (1H, m);
H: 1.48 s (9H), 1.68 (3H, t, J 3.1), 3.72 (4H, br. s), 4.65 (2H, m), 5.12 (2H, m), 5.75 (1H, m);  C: 15.91+, 28.34+, 48.74-, 75.23-, 79.53, 95.62, 116.20-, 116.63-, 133.76+, 155.52, 206.44; n
max: 2978 m, 2929 m, 1698 s, 1456 m, 1407 s, 1366 m, 1244 s, 1167 s, cm-1; N-allyl-N-t-butoxycarbonyl-N-(2-bromo-1-methylcyclopropylmethyl)amine (20a) (188 mg, 0.62 mmole, 14 %) which showed
C: 15.91+, 28.34+, 48.74-, 75.23-, 79.53, 95.62, 116.20-, 116.63-, 133.76+, 155.52, 206.44; n
max: 2978 m, 2929 m, 1698 s, 1456 m, 1407 s, 1366 m, 1244 s, 1167 s, cm-1; N-allyl-N-t-butoxycarbonyl-N-(2-bromo-1-methylcyclopropylmethyl)amine (20a) (188 mg, 0.62 mmole, 14 %) which showed  H: 0.69 (1H, dd, J 6.4, 4.5 Hz), 0.87 (1H, dd, J 7.4, 6.4 Hz), 1.25 (3H, s), 1.47 (9H, s), 2.95 (1H, br. s), 3.30 - 4.00 (4H, br. m), 5.07 - 5.17 (2H, m), 5.74 (1H, ddt, J 17.0, 10.5, 5.6 Hz); n
max: 3079 w, 2977 m, 2930 m, 1694 s, 1456 m, 1410 m, 1366 m, 1246 m, 1172 s cm-1.
H: 0.69 (1H, dd, J 6.4, 4.5 Hz), 0.87 (1H, dd, J 7.4, 6.4 Hz), 1.25 (3H, s), 1.47 (9H, s), 2.95 (1H, br. s), 3.30 - 4.00 (4H, br. m), 5.07 - 5.17 (2H, m), 5.74 (1H, ddt, J 17.0, 10.5, 5.6 Hz); n
max: 3079 w, 2977 m, 2930 m, 1694 s, 1456 m, 1410 m, 1366 m, 1246 m, 1172 s cm-1.
(1R, 4S, 5S)-N-t-Butoxycarbonyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane.
A 1.5M solution of methyllithium in ether (0.44 ml, 0.66 mmole) was added to (S)-(+)-N-t-butoxy-carbonyl-N-benzyl-2,2-dibromo-1-methylcyclopropylmethylamine (260 mg, 0.60 mmole) in dry ether (10 ml) under nitrogen at 20 o
C over 5 min. The solution was stirred 30 min then sat.aq. ammonium chloride (5 ml) was added. The water layer was extracted with ether (10 ml). Evaporation of the combined organic layers and chromatography over silica (ether-petrol, 3:17) gave (1R, 4S, 5S)-N-t-butoxycarbonyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (18b), (65 mg, 0.24 mmole, 40 %) as a mixture of two rotamers in ratio 2:1, [ ]D20-98.2o
(c 0.708, CHCl3) which showed n
max: 2978 m, 2929 m, 2868 m, 2250 m, 1684 s, 1478 m, 1455 m, 1408 s, 1390 s, 1367 m, 1172 s, 1127 s, 908 s, 879 m, 731 s, 699 m, 649 m cm-1; major rotamer,
]D20-98.2o
(c 0.708, CHCl3) which showed n
max: 2978 m, 2929 m, 2868 m, 2250 m, 1684 s, 1478 m, 1455 m, 1408 s, 1390 s, 1367 m, 1172 s, 1127 s, 908 s, 879 m, 731 s, 699 m, 649 m cm-1; major rotamer,  H: 0.52 (1H, m), 0.69 (1H, m), 1.25 (1H, m), 1.27 (9H, s), 1.35 (3H, s), 3.44 (1H, d, J 10.7 Hz), 3.75 (1H, d, J 10.7 Hz), 4.69 (1H, s), 7.19 - 7.36 (5H, m);
H: 0.52 (1H, m), 0.69 (1H, m), 1.25 (1H, m), 1.27 (9H, s), 1.35 (3H, s), 3.44 (1H, d, J 10.7 Hz), 3.75 (1H, d, J 10.7 Hz), 4.69 (1H, s), 7.19 - 7.36 (5H, m);  C: 16.80-, 18.77+, 22.91, 28.28+, 30.14+, 53.26-, 64.32+, 79.38, 126.54+, 128.28+, 143.85, 154.56; minor rotamer,
C: 16.80-, 18.77+, 22.91, 28.28+, 30.14+, 53.26-, 64.32+, 79.38, 126.54+, 128.28+, 143.85, 154.56; minor rotamer,  H: 0.52 (1H, m), 0.69 (1H, m), 1.25 (1H, m), 1.32 (3H, s), 1.41 (9H, s), 3.37 (1H, d, J 10.8 Hz), 3.69 (1H, d, J 10.8 Hz), 4.89 (1H, s), 7.19 - 7.36 (5H, m);
H: 0.52 (1H, m), 0.69 (1H, m), 1.25 (1H, m), 1.32 (3H, s), 1.41 (9H, s), 3.37 (1H, d, J 10.8 Hz), 3.69 (1H, d, J 10.8 Hz), 4.89 (1H, s), 7.19 - 7.36 (5H, m);  C: 16.35-, 18.56+, 23.49, 28.28+, 29.19+, 53.46-, 63.45+, 79.38, 126.97+, 128.47+, 143.17, 154.33.
C: 16.35-, 18.56+, 23.49, 28.28+, 29.19+, 53.46-, 63.45+, 79.38, 126.97+, 128.47+, 143.17, 154.33.
Also was isolated a mixture of (19b), (20b) and starting material (90 mg, 27:7:11 based on 1H NMR of Boc-deprotected mixture) which showed n
max: 3030 m, 2976 s, 2930 s, 1961 m, 1694 s, 1496 m, 1454 s, 1413 s, 1365 s, 1242 s, 1169 s, 1124 s, 877 m, 768 m, 699 s cm-1; N-benzyl-N-t-butoxycarbonyl-N-2-methylbuta-2,3-dienylamine (19b) showed (from this mixture)  H: 1.51 (9H, m), 1.68 (3H, m), 3.80 (2H, m), 4.45 (2H, m), 4.73 (2H, m), 7.21-7.39 (5H, m);
H: 1.51 (9H, m), 1.68 (3H, m), 3.80 (2H, m), 4.45 (2H, m), 4.73 (2H, m), 7.21-7.39 (5H, m);  C: 15.95, 28.39, 48.62, 75.37, 95.41, 127.09, 128.36, 128.58, 137.53, 156.31, 206.83. This mixture (90 mg) was deprotected by reaction with trifluoroacetic acid to give 69 mg of amines which were separated by column chromatography to yield N-benzyl-N-2-methylbuta-2,3-dienylamine (31 mg, 0.18 mmole, 27 %) which showed
C: 15.95, 28.39, 48.62, 75.37, 95.41, 127.09, 128.36, 128.58, 137.53, 156.31, 206.83. This mixture (90 mg) was deprotected by reaction with trifluoroacetic acid to give 69 mg of amines which were separated by column chromatography to yield N-benzyl-N-2-methylbuta-2,3-dienylamine (31 mg, 0.18 mmole, 27 %) which showed  H: 1.70 (3H, t, J 3.1 Hz), 3.20 (2H, t, J 3.0 Hz), 3.27 (1H, br. s), 3.85 (2H, s), 4.76 (2H, qt, J 3.1, 3.0 Hz), 7.25 - 7.34 (5H, m); n
max: 1960 s cm-1; N-benzyl-N-(2-bromo-1-methylcyclopropylmethyl)amine (11 mg, 0.045 mmole, 7 %) which showed
H: 1.70 (3H, t, J 3.1 Hz), 3.20 (2H, t, J 3.0 Hz), 3.27 (1H, br. s), 3.85 (2H, s), 4.76 (2H, qt, J 3.1, 3.0 Hz), 7.25 - 7.34 (5H, m); n
max: 1960 s cm-1; N-benzyl-N-(2-bromo-1-methylcyclopropylmethyl)amine (11 mg, 0.045 mmole, 7 %) which showed  H: 0.68 (1H, dd, J 6.4, 4.3 Hz), 1.04 (1H, dd, J 8.1, 6.4 Hz), 1.33 (3H, s), 1.55 (1H, s), 2.46 (1H, d, J 12.3 Hz), 2.62 (1H, d, J 12.3 Hz), 2.89 (1H, dd, J 8.1, 4.3 Hz), 3.79 (2H, s), 7-24 - 7.32 (5H, m); (m/z, %): 255, 5; 254, 7; 253, 5 (M+); 252, 7; 174, 12; 160, 13; 120, 41; 91, 100; 65, 14.
H: 0.68 (1H, dd, J 6.4, 4.3 Hz), 1.04 (1H, dd, J 8.1, 6.4 Hz), 1.33 (3H, s), 1.55 (1H, s), 2.46 (1H, d, J 12.3 Hz), 2.62 (1H, d, J 12.3 Hz), 2.89 (1H, dd, J 8.1, 4.3 Hz), 3.79 (2H, s), 7-24 - 7.32 (5H, m); (m/z, %): 255, 5; 254, 7; 253, 5 (M+); 252, 7; 174, 12; 160, 13; 120, 41; 91, 100; 65, 14.
(1R, 4S, 5S)-1-Methyl-4-vinyl-3-azabicyclo[3.1.0]hexane.
(1R, 4S, 5S)-N-t-Butoxycarbonyl-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane (30.0 mg, 0.134 mmole) was dissolved in dichloromethane (0.5 ml) and trifluoroacetic acid (0.5 ml) was added and the solution was stirred for 30 min. The volatiles were removed in vacuo and 1 % aq. sodium hydroxide (2 ml, 0.5 mmole) was added and the product was extracted with chloroform (2 x 2 ml). The combined organic layers were washed with 1 % aq. sodium hydroxide (2 ml) and then dried. The solvent was removed to yield (1R, 4S, 5S)-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane (21a), (15.8 mg, 0.128 mmole, 96 %), (Found: M+, 123.1050. Calculated for C8H13N: 123.1048), [ ]D20-5.4o
(c 0.67, CHCl3) which showed
]D20-5.4o
(c 0.67, CHCl3) which showed  H: 0.42 - 0.50 (2H, m), 1.05 (1H, dd, J 7.3, 4.4 Hz), 1.26 (3H, s), 2.02 (1H, s), 2.81 (2H, s), 3.52 (1H, d, J 6.7 Hz), 5.02 (1H, ddd, J 10.3, 1.8, 1.0 Hz), 5.13 (1H, ddd, J 17.0, 1.8, 1.3), 5.87 (1H, ddd, J 17.0, 10.3, 6.7 Hz);
H: 0.42 - 0.50 (2H, m), 1.05 (1H, dd, J 7.3, 4.4 Hz), 1.26 (3H, s), 2.02 (1H, s), 2.81 (2H, s), 3.52 (1H, d, J 6.7 Hz), 5.02 (1H, ddd, J 10.3, 1.8, 1.0 Hz), 5.13 (1H, ddd, J 17.0, 1.8, 1.3), 5.87 (1H, ddd, J 17.0, 10.3, 6.7 Hz);  C: 13.60-, 18.39+, 24.35, 28.23+, 52.17-, 62.10+, 113.36-, 140.73+; n
max: 3318 br. s, 3079 m, 2943 s, 2866 s, 1639 m, 1450 m, 1423 m, 1387 w, 1101 m, 1019 m, 916 s, 890 m cm-1; (m/z, %): 124, 10; 123,48 (M+); 122, 25; 108, 49; 96, 100; 94, 37; 80, 34.
C: 13.60-, 18.39+, 24.35, 28.23+, 52.17-, 62.10+, 113.36-, 140.73+; n
max: 3318 br. s, 3079 m, 2943 s, 2866 s, 1639 m, 1450 m, 1423 m, 1387 w, 1101 m, 1019 m, 916 s, 890 m cm-1; (m/z, %): 124, 10; 123,48 (M+); 122, 25; 108, 49; 96, 100; 94, 37; 80, 34.
(1R, 4S, 5S)-1-Methyl-4-phenyl-3-azabicyclo[3.1.0]hexane.
(1R, 4S, 5S)-N-t-Butoxycarbonyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (25 mg, 0.091 mmole) was dissolved in dichloromethane (0.5 ml) and trifluoroacetic acid (0.5 ml) was added and the solution was stirred for 30 min. The volatiles were removed in vacuo and 1 % aq. sodium hydroxide (2 ml, 0.5 mmole) was added and the product was extracted with chloroform (2 x 2 ml). The combined organic layers were washed with 1 % aq. sodium hydroxide (2 ml) and then dried. The solvent was removed to yield (1R, 4S, 5S)-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (21b) (14 mg, 0.081 mmole, 89 %), (Found: M+, 173.1205. calculated for C8H13N: 173.1205), m.p. 34 - 35 o
C, [ ]D20-10.6o
(c 0.81, CHCl3) which showed
]D20-10.6o
(c 0.81, CHCl3) which showed  H: 0.59 - 0.64 (2H, m), 1.28 (1H, dd, J 7.1, 4.9 Hz), 1.34 (3H, s), 2.81 (2H, s), 4.20 (1H, s), 4.32 (1H, br.s), 7.22 - 7.37 (5H, m);
H: 0.59 - 0.64 (2H, m), 1.28 (1H, dd, J 7.1, 4.9 Hz), 1.34 (3H, s), 2.81 (2H, s), 4.20 (1H, s), 4.32 (1H, br.s), 7.22 - 7.37 (5H, m);  C: 14.65-, 18.15+, 25.18, 29.30+, 51.77-, 64.09+, 126.96+, 127.28+, 128.65+, 143.46; n
max: 3313 br. m, 3060 m, 3027 m, 2985 m, 2941 s, 2865 s, 1492 m, 1453 s, 1408 m, 1387 m, 1370 m, 1101 m, 1028 m, 880 m, 755 m, 700 s cm-1; (m/z, %): 174, 44; 173, 58 (M+); 172, 56; 158, 27; 149, 29; 117, 55; 91, 100; 83, 39; 57, 44.
C: 14.65-, 18.15+, 25.18, 29.30+, 51.77-, 64.09+, 126.96+, 127.28+, 128.65+, 143.46; n
max: 3313 br. m, 3060 m, 3027 m, 2985 m, 2941 s, 2865 s, 1492 m, 1453 s, 1408 m, 1387 m, 1370 m, 1101 m, 1028 m, 880 m, 755 m, 700 s cm-1; (m/z, %): 174, 44; 173, 58 (M+); 172, 56; 158, 27; 149, 29; 117, 55; 91, 100; 83, 39; 57, 44.
(1R, 4S, 5S)-N-t-Butoxycarbonyl-1-methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid.
(1R, 4S, 5S)-N-t-Butoxycarbonyl-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane (200 mg, 0.896 mmole) was dissolved in carbon tetrachloride (2 ml) and acetonitrile (2 ml), water (3 ml), periodic acid (1.07 g, 4.7 mmole, 5.25 eq.) and ruthenium trichloride hydrate (4.0 mg, 0.02 eq.) were added. The reaction mixture was vigorously stirred for 3 h at room temperature, then ether (5 ml) was added. The black organic solution was separated, and the water layer was washed with ether (2 x 5 ml). The ether solutions were combined, dried twice with magnesium sulfate with vigorous shaking (until all colour went into the solid phase and the solution became colourless and trans-parent) and the solvent was removed in vacuo to give (1R, 4S, 5S)-N-t-butoxycarbonyl-1-methyl-3-azabicyclo-[3.1.0]hexane-4-carboxylic acid (22) (195 mg, 0.808 mmole, 90 %) as a mixture of two rotamers (1:1), m.p. 117 - 119 o
C, [ ]D20-86.3o
(c 0.730, CHCl3) which showed
]D20-86.3o
(c 0.730, CHCl3) which showed  H: 0.51 (1H, dd, J 5.2, 3.8 Hz), 0.55 (1H, dd, J 5.2, 3.8 Hz), 0.71 (2H, dd, J 7.6, 5.2 Hz), 1.27 (3H, s), 1.28 (3H, s), 1.43 (9H, s), 1.48 (9H, s), 1.50 (2H, m), 3.37 (1H, d, J 10.3 Hz), 3.42 (1H, d, J 10.2 Hz), 3.59 (1H, d, J 10.3 Hz), 3.64 (1H, d, J 10.2 Hz), 4.27 (1H, s), 4.38 (1H, s), 9.85 (2H, s);
H: 0.51 (1H, dd, J 5.2, 3.8 Hz), 0.55 (1H, dd, J 5.2, 3.8 Hz), 0.71 (2H, dd, J 7.6, 5.2 Hz), 1.27 (3H, s), 1.28 (3H, s), 1.43 (9H, s), 1.48 (9H, s), 1.50 (2H, m), 3.37 (1H, d, J 10.3 Hz), 3.42 (1H, d, J 10.2 Hz), 3.59 (1H, d, J 10.3 Hz), 3.64 (1H, d, J 10.2 Hz), 4.27 (1H, s), 4.38 (1H, s), 9.85 (2H, s);  C: 15.75-, 16.00-, 18.17+, 18.26+, 22.51, 23.14, 25.13+, 26.14+, 28.23+, 28.35+, 53.07-, 53.36-, 61.65+, 62.06+, 80.44, 80.68, 154.59, 155.54, 176.89, 177.90; nmax (CCl4): 3150 br. m, 2972 s, 2951 s, 2881 s, 1736 s, 1636 s, 1478 m, 1458 s, 1432 s, 1407 m, 1368 m, 1326 m, 1248 s, 1214 m, 1191 s, 1167 s, 1135 s, 898 m cm-1.
C: 15.75-, 16.00-, 18.17+, 18.26+, 22.51, 23.14, 25.13+, 26.14+, 28.23+, 28.35+, 53.07-, 53.36-, 61.65+, 62.06+, 80.44, 80.68, 154.59, 155.54, 176.89, 177.90; nmax (CCl4): 3150 br. m, 2972 s, 2951 s, 2881 s, 1736 s, 1636 s, 1478 m, 1458 s, 1432 s, 1407 m, 1368 m, 1326 m, 1248 s, 1214 m, 1191 s, 1167 s, 1135 s, 898 m cm-1.
(1R, 4S, 5S)-1-Methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid.
(1R, 4S, 5S)-N-t-Butoxycarbonyl-1-methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid (163 mg, 0.676 mmole) was dissolved in dichloromethane (2 ml) and trifluoroacetic acid (2 ml) was added and the solution was stirred for 2 h. The volatiles were removed in vacuo and the residue was dissolved in water and desalted on a column of Dowex 50W - X8 resin in the hydrogen form. The amino acid was eluted from the column with 5M ammonia. After evaporation in vacuo and drying over phosphorus pentoxide, (1R, 4S, 5S)-1-methyl-3-azabicyclo-[3.1.0]hexane-4-carboxylic acid was obtained (23) (85 mg, 0.606 mmole, 89 %), m.p. 236 - 240 o
C (decomp.), [ ]D20-79o
(c 0.758, H2O) which showed
]D20-79o
(c 0.758, H2O) which showed  H (D2O): 0.59 (1H, dd, J 6.4, 4.2 Hz), 0.89 (1H, dd, J 8.3, 6.4 Hz), 1.29 (3H, s), 1.75 (1H, dd, J 8.3, 4.2 Hz), 3.39 (1H, d, J 11.2 Hz), 3.44 (1H, d, J 11.2 Hz), 4.08 (1H, s);
H (D2O): 0.59 (1H, dd, J 6.4, 4.2 Hz), 0.89 (1H, dd, J 8.3, 6.4 Hz), 1.29 (3H, s), 1.75 (1H, dd, J 8.3, 4.2 Hz), 3.39 (1H, d, J 11.2 Hz), 3.44 (1H, d, J 11.2 Hz), 4.08 (1H, s);  C (D2O): 16.71-, 19.30+, 25.26, 28.06+, 53.80-, 66.45+, 176.45; n
max (KBr): 3420 br. w, 3264 w, 2944 w, 2300 - 2800 br. w, 1616 s, 1376 s, 1312 m, 1275 w, 1202 m, 669 w cm-1.
C (D2O): 16.71-, 19.30+, 25.26, 28.06+, 53.80-, 66.45+, 176.45; n
max (KBr): 3420 br. w, 3264 w, 2944 w, 2300 - 2800 br. w, 1616 s, 1376 s, 1312 m, 1275 w, 1202 m, 669 w cm-1.
Ozonolysis of (1R, 4S, 5S)-N-t-butoxycarbonyl-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane.
Ozone (5 % in oxygen) was passed through a solution of (1R, 4S, 5S)-N-t-butoxy-carbonyl-1-methyl-4-vinyl-3-azabicyclo[3.1.0]hexane (31.5 mg, 0.141 mmole) in acetone (10 ml) at -78 o
C for 5 min and the reaction mixture was kept at -78 o
C for 30 min, then allowed to warm to 0 o
C and Jones' reagent (0.05 g of CrO3 in 1 ml of 15 % sulfuric acid) was added. After 1 h at room temperature chromium salts were filtered off and acetone was removed in vacuo. The residue was dissolved in ether (5 ml) and this solution was extracted with 0.1M sodium hydroxide (5 ml, 0.5 mmole). The organic layer was dried with magnesium sulfate to give after evaporation of the solvent (1R, 5S)-N-t-butoxycarbonyl-1-methyl-4-oxo-3-azabicyclo-[3.1.0]hexane (24) (13.7 mg, 0.065 mmole, 46 %) which showed  H: 0.96 (1H, dd, J 4.7, 3.1 Hz), 1.12 (1H, dd, J 8.7, 4.7 Hz), 1.37 (3H, s), 1.53 (9H, s), 1.81 (1H, dd, J 8.7, 3.1 Hz), 3.57 (1H, d, J 10.9 Hz), 3.76 (1H, d, J 10.9 Hz);
H: 0.96 (1H, dd, J 4.7, 3.1 Hz), 1.12 (1H, dd, J 8.7, 4.7 Hz), 1.37 (3H, s), 1.53 (9H, s), 1.81 (1H, dd, J 8.7, 3.1 Hz), 3.57 (1H, d, J 10.9 Hz), 3.76 (1H, d, J 10.9 Hz);  C: 18.62+, 19.31-, 27.74, 28.03+, 59.92-, 82.52, 150.29, 174.12; n
max (CCl4): 2978 s, 2932 s, 1789 s, 1748 s, 1714 s, 1480 m, 1454 m, 1369 s, 1351 s, 1322 s, 1293 s, 1258 m, 1159 s, 1091 m, 1056 m, 1007 m, 913 m, 856 m, 781 m cm-1. Hydrochloric acid (1 ml, 1M, 1 mmole) was added to the water layer and this was extracted with ether (2 x 5 ml). The organic fractions were combined and dried to give after evaporation of the solvent (1R, 4S, 5S)-N-t-butoxycarbonyl-1-methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid (22) (14.0 mg, 0.058 mmole, 41 %), identical to that above.
C: 18.62+, 19.31-, 27.74, 28.03+, 59.92-, 82.52, 150.29, 174.12; n
max (CCl4): 2978 s, 2932 s, 1789 s, 1748 s, 1714 s, 1480 m, 1454 m, 1369 s, 1351 s, 1322 s, 1293 s, 1258 m, 1159 s, 1091 m, 1056 m, 1007 m, 913 m, 856 m, 781 m cm-1. Hydrochloric acid (1 ml, 1M, 1 mmole) was added to the water layer and this was extracted with ether (2 x 5 ml). The organic fractions were combined and dried to give after evaporation of the solvent (1R, 4S, 5S)-N-t-butoxycarbonyl-1-methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid (22) (14.0 mg, 0.058 mmole, 41 %), identical to that above.
(S)-(-)-N,N-Dibenzyl-2,2-dibromo-1-methylcyclopropylmethylamine.
A solution of sodium carbonate (1.06 g, 10 mmole) in water (5 ml) was added to a solution of (S)-(-)-N-benzyl-2,2-dibromo-1-methylcyclopropylmethylamine (1.53 g, 4.59 mmole) and benzylbromide (1.03 g, 6.0 mmole) in dichloromethane (10 ml) and the mixture was refluxed and stirred for 2 days. The water layer was extracted with dichloromethane (10 ml). The combined organic layers were dried and solvent was removed. The residue was dissolved in ethanol (15 ml) and sodium hydroxide (0.2 g, 5 mmole) was added. The mixture was refluxed for 1 h, the solvent was removed and water (10 ml) and petrol (15 ml) were added. The water layer was extracted with petrol (15 ml). The combined organic layers were dried and solvent was removed. The crude product was purified by column chromatography over silica to give (S)-(-)-N,N-dibenzyl-2,2-dibromo-1-methylcyclopropylmethylamine (25), (1.81 g, 4.27 mmole, 93 %), (Found: C 54.05, H 5.05, N 3.37. Calculated for C19H21Br2N: C 53.93, H 5.00, N 3.31), m.p. 55-56 o
C, [ ]D20-21.6o
(c 0.984, CHCl3) which showed
]D20-21.6o
(c 0.984, CHCl3) which showed  H: 1.42 (1H, d, J 7.4 Hz), 1.45 (1H, d, J 7.4 Hz), 1.51 (3H, s), 2.63 (1H, d, J 13.0 Hz), 2.70 (1H, d, J 13.0 Hz), 3.53 (2H, d, J 13.6 Hz), 3.69 (2H, d, J 13.6 Hz), 7.23-7.44 (10H, m);
H: 1.42 (1H, d, J 7.4 Hz), 1.45 (1H, d, J 7.4 Hz), 1.51 (3H, s), 2.63 (1H, d, J 13.0 Hz), 2.70 (1H, d, J 13.0 Hz), 3.53 (2H, d, J 13.6 Hz), 3.69 (2H, d, J 13.6 Hz), 7.23-7.44 (10H, m);  C: 22.41+, 28.22, 33.58-, 38.71, 58.09-, 60.23-, 127.02+, 128.36+, 128.75+, 139.62; n
max: 3084 m, 3062 m, 3027 s, 2989 m, 2966 m, 2928 s, 28 86 m, 2828 s, 2797 s, 1601 m, 1494 s, 1453 s, 1427 m, 1384 m, 1367 s, 1323 m, 1239 m, 1126 s, 1069 s, 1027 s, 980 s, 917 m, 905 m, 745 s, 696 s cm-1.
C: 22.41+, 28.22, 33.58-, 38.71, 58.09-, 60.23-, 127.02+, 128.36+, 128.75+, 139.62; n
max: 3084 m, 3062 m, 3027 s, 2989 m, 2966 m, 2928 s, 28 86 m, 2828 s, 2797 s, 1601 m, 1494 s, 1453 s, 1427 m, 1384 m, 1367 s, 1323 m, 1239 m, 1126 s, 1069 s, 1027 s, 980 s, 917 m, 905 m, 745 s, 696 s cm-1.
(1R, 4R, 5S)-N-Benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane.
a) A 1.5M solution of methyllithium in ether (1.75 ml, 2.63 mmole) was added to a solution of (S)-(-)-N,N-dibenzyl-2,2-dibromo-1-methylcyclopropylmethylamine (1.06 g, 2.50 mmole) in dry ether (25 ml) under nitrogen at - 90 o
C for 5 min. The solution was stirred for 30 min at - 90 o
C then warmed to 0 o
C for 30 min and water (5 ml) was added. The water layer was extracted with ether (10 ml). The combined organic layers were evaporated and purified by chromatography over silica (ether - petrol, 1:24) to give crude (>90 % purity by NMR) (1R, 4R, 5S)- N-benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (26), (0.63 g, 2.39 mmole, 96 %), and pure (1R, 4S, 5S)- N-benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (27), (12 mg, 0.046 mmole, 1.8 %), (Found: M+, 263.1674. Calculated for C19H21N: 263.1674), [ ]D20-25.4o
(c 0.67, CHCl3) which showed
]D20-25.4o
(c 0.67, CHCl3) which showed  H: 0.56 (1H, dd, J 7.7, 3.8 Hz), 1.12 (1H, dd, J 3.8, 3.6 Hz), 1.21 (1H, dd, J 7.7, 3.6 Hz), 1.39 (3H, s), 2.64 (1H, d, J 8.7 Hz), 2.83 (1H, d, J 8.7 Hz), 3.18 (1H, d, J 14.0 Hz), 3.45 (1H, d, J 14.0 Hz), 4.04 (1H, s), 7.09 - 7.51 (10H, m);
H: 0.56 (1H, dd, J 7.7, 3.8 Hz), 1.12 (1H, dd, J 3.8, 3.6 Hz), 1.21 (1H, dd, J 7.7, 3.6 Hz), 1.39 (3H, s), 2.64 (1H, d, J 8.7 Hz), 2.83 (1H, d, J 8.7 Hz), 3.18 (1H, d, J 14.0 Hz), 3.45 (1H, d, J 14.0 Hz), 4.04 (1H, s), 7.09 - 7.51 (10H, m);  C: 16.22, 19.03, 23.30, 29.35, 54.08, 57.21, 67.38, 126.43, 126.88, 127.99, 128.05, 128.15, 128.55, 140.40, 141.99; n
max: 3062 m, 3028 m, 2924 s, 2866 s, 2797 m, 1696 m, 1493 m, 1453 s, 1366 m, 1168 m, 1028 m, 762 m, 732 m, 700 s cm-1; (m\z, %): 264, 9; 263, 45 (M+); 262, 23; 187, 20; 186, 76; 172, 25; 91, 100.
C: 16.22, 19.03, 23.30, 29.35, 54.08, 57.21, 67.38, 126.43, 126.88, 127.99, 128.05, 128.15, 128.55, 140.40, 141.99; n
max: 3062 m, 3028 m, 2924 s, 2866 s, 2797 m, 1696 m, 1493 m, 1453 s, 1366 m, 1168 m, 1028 m, 762 m, 732 m, 700 s cm-1; (m\z, %): 264, 9; 263, 45 (M+); 262, 23; 187, 20; 186, 76; 172, 25; 91, 100.
Reaction of (S)-(-)-N,N-dibenzyl-2,2-dibromo-1-methylcyclopropylmethylamine (212 mg, 0.50 mmole) with methyl lithium (0.525 mmole) as above but at room temperature, with work up and column chromatography as before gave (27) (6.7 mg, 0.025 mmole, 5 %) identical to that above and a mixture of N-Benzyl-N-t-butoxycarbo-nyl-N-2-methylbuta-2,3-dienylamine (28) with the major compound (26) (1:2, 117 mg, 0.445 mmole, 89 %). By subtraction of the spectra of pure (26) from the spectra of this mixture compound (28) showed  H: 1.85 (3H, t, J 3.1 Hz), 3.07 (2H, t, J 3.0 Hz), 3.70 (4H, s), 4.76 (2H, qt, J 3.1, 3.0 Hz), 7.25 - 7.64 (10H, m);
H: 1.85 (3H, t, J 3.1 Hz), 3.07 (2H, t, J 3.0 Hz), 3.70 (4H, s), 4.76 (2H, qt, J 3.1, 3.0 Hz), 7.25 - 7.64 (10H, m);  C: 16.92+, 57.37-, 57.92-, 73.78-, 96.64, 126.87+, 128.24+, 128.87+, 139.75, 207.78; n
max: 1956 m cm-1.
C: 16.92+, 57.37-, 57.92-, 73.78-, 96.64, 126.87+, 128.24+, 128.87+, 139.75, 207.78; n
max: 1956 m cm-1.
b) Crude (1R, 4R, 5S)-N-benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (52.5 mg, 0.200 mmole) was dissolved in ethanol (2.5 ml) and 3M hydrochloric acid (0.13 ml, 0.40 mmole) and 5 % Pd/C (5 mg) were added. After stirring for 1 h under hydrogen the reaction mixture was filtered, ethanol was evaporated and 1 % aq. sodium hydroxide (2.5 ml, 0.6 mmole) was added. The amine was extracted with ether (2 x 5 ml), the organic solution was dried, evaporated and the product was passed though a small pad of silica (ether - petrol, 4:96) to give pure (1R, 4R, 5S)-N-benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (26), (43.6 mg, 0.166 mmole, 83 %), (Found: M+, 263.1674. Calculated for C19H21N: 263.1674), [ ]D20 +35.8o
(c 0.70, CHCl3) which showed
]D20 +35.8o
(c 0.70, CHCl3) which showed  H: 0.14 (1H, dd, J 7.6, 4.0 Hz), 1.20 (4H, s), 1.33 (1H, m), 2.28 (1H, d, J 8.6 Hz), 3.02 (1H, d, J 8.6 Hz), 3.10 (1H, d, J 13.3 Hz), 3.79 (1H, d, J 13.3 Hz), 3.79 (1H, d, J 3.7 Hz), 7.26 - 7.50 (10H, m);
H: 0.14 (1H, dd, J 7.6, 4.0 Hz), 1.20 (4H, s), 1.33 (1H, m), 2.28 (1H, d, J 8.6 Hz), 3.02 (1H, d, J 8.6 Hz), 3.10 (1H, d, J 13.3 Hz), 3.79 (1H, d, J 13.3 Hz), 3.79 (1H, d, J 3.7 Hz), 7.26 - 7.50 (10H, m);  C: 11.81-, 19.11+, 21.59, 30.94+, 56.94-, 59.73-, 68.71+, 126.64+, 127.01+, 127.41+, 128.05+, 128.31+, 128.45+, 139.72, 142.37; n
max: 3064 s, 3027 s, 2997 m, 2944 s, 2894 s, 2787 s, 1602 m, 1494 s, 1452 s, 1371 s, 1351 m, 1294 m, 1234 m, 1196 m, 1165 m, 1132 m, 1070 m, 1028 s, 963 m, 911 m, 751 s, 698 s cm-1; (m\z, %): 264, 13; 263, 68 (M+); 262, 66; 187, 11; 186, 56; 172, 32; 91, 100; 65, 25.
C: 11.81-, 19.11+, 21.59, 30.94+, 56.94-, 59.73-, 68.71+, 126.64+, 127.01+, 127.41+, 128.05+, 128.31+, 128.45+, 139.72, 142.37; n
max: 3064 s, 3027 s, 2997 m, 2944 s, 2894 s, 2787 s, 1602 m, 1494 s, 1452 s, 1371 s, 1351 m, 1294 m, 1234 m, 1196 m, 1165 m, 1132 m, 1070 m, 1028 s, 963 m, 911 m, 751 s, 698 s cm-1; (m\z, %): 264, 13; 263, 68 (M+); 262, 66; 187, 11; 186, 56; 172, 32; 91, 100; 65, 25.
(1R, 4R, 5S)-1-Methyl-4-phenyl-3-azabicyclo[3.1.0]hexane.
a) Crude (1R, 4R, 5S)-N-benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (620 mg, 2.35 mmole) was dissolved in ethanol (6 ml) and 3M hydrochloric acid (1.57 ml, 4.70 mmole) and 5 % Pd/C (310 mg) were added. After stirring for 18 h under hydrogen, the reaction mixture was filtered, ethanol was evaporated and 1 % aq. sodium hydroxide (25 ml, 6.0 mmole) was added. The amine was extracted with ether (2 x 50 ml), the organic solution was dried and evaporated to give crude (1R, 4R, 5S)-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (30) (425 mg, >80 % purity by glc and PMR).
b) (1R, 4R, 5S)-N-benzyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (132 mg, 0.50 mmole) was dis-solved in ethanol (5 ml) and 3M hydrochloric acid (0.33 ml, 1.00 mmole) and 5 % Pd/C (66 mg) were added. After stirring for 18 h under hydrogen, the reaction mixture was filtered, ethanol was evaporated and 1 % aq. sodium hydroxide (5 ml, 1.20 mmole) was added. The amine was extracted with ether (2 x 10 ml), the organic solution was dried and evaporated to give (1R, 4R, 5S)-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (30) (82 mg, 0.473 mmole, 95 %), (Found: M+, 173.1190. Calculated for C12H15N: 173.1204), [ ]D20 +146.4o
(c 1.070, CHCl3) which showed
]D20 +146.4o
(c 1.070, CHCl3) which showed  H: 0.48 (1H, dd, J 7.7, 5.0 Hz), 0.69 (1H, dd, J 5.0, 3.9 Hz), 1.33 (3H, s), 1.39 (1H, ddd, J 7.7, 3.9, 3.1 Hz), 1.79 (1H, s), 2.95 (1H, d, J 10.8 Hz), 3.06 (1H, d, J 10.8 Hz), 4.66 (1H, d, J 3.1 Hz), 7.22 - 7.50 (5H,m);
H: 0.48 (1H, dd, J 7.7, 5.0 Hz), 0.69 (1H, dd, J 5.0, 3.9 Hz), 1.33 (3H, s), 1.39 (1H, ddd, J 7.7, 3.9, 3.1 Hz), 1.79 (1H, s), 2.95 (1H, d, J 10.8 Hz), 3.06 (1H, d, J 10.8 Hz), 4.66 (1H, d, J 3.1 Hz), 7.22 - 7.50 (5H,m);  C: 10.68-, 18.47+, 24.28, 28.24+, 54.62-, 62.80+, 126.99+, 127.13+, 128.20+, 142.04; n
max: 3322 br. m, 3060 m, 3028 m, 2993 m, 2941 s, 2893 s, 2864 s, 1494 m, 1451 s, 1418 m, 1388 m, 1347 m, 1289 m, 1215 m, 1195 m, 1087 m, 1029 m, 963 m, 897 m, 750 s, 700s cm-1.
C: 10.68-, 18.47+, 24.28, 28.24+, 54.62-, 62.80+, 126.99+, 127.13+, 128.20+, 142.04; n
max: 3322 br. m, 3060 m, 3028 m, 2993 m, 2941 s, 2893 s, 2864 s, 1494 m, 1451 s, 1418 m, 1388 m, 1347 m, 1289 m, 1215 m, 1195 m, 1087 m, 1029 m, 963 m, 897 m, 750 s, 700s cm-1.
(1R, 4R, 5S)-1-Methyl-4-phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane.
(1R, 4R, 5S)-1-Methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (65 mg, 0.375 mmole) was dissolved in dichloromethane (5 ml) and trifluoroacetic anhydride (1 ml, 5 mmole) was added. The solution was stirred for 2 h, the volatiles were removed in vacuo and the residue was dissolved in chloroform (5 ml). This solution was extracted with phosphate buffer (2 x 5 ml, pH 6.88), dried and, after removing of the solvent, (1R, 4R, 5S)-1-methyl-4-phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane (31) (99 mg, 0.367 mmole, 98 %) was obtained, m.p. 93 - 95 o
C, [ ]D20 +164.3o
(c 0.890, CHCl3) which showed
]D20 +164.3o
(c 0.890, CHCl3) which showed  H: 0.55 (1H, dd, J 7.6, 5.1 Hz), 0.63 (1H, dd, J 5.1, 4.7 Hz), 1.35 (3H, s), 1.74 (1H, ddd, J 7.6, 5.1, 4.7 Hz), 3.84 (1H, d, J 10.4 Hz), 4.05 (1H, d, J 10.4 Hz), 5.37 (1H, d, J 5.1 Hz), 7.05 - 7.36 (5H, m);
H: 0.55 (1H, dd, J 7.6, 5.1 Hz), 0.63 (1H, dd, J 5.1, 4.7 Hz), 1.35 (3H, s), 1.74 (1H, ddd, J 7.6, 5.1, 4.7 Hz), 3.84 (1H, d, J 10.4 Hz), 4.05 (1H, d, J 10.4 Hz), 5.37 (1H, d, J 5.1 Hz), 7.05 - 7.36 (5H, m);  C: 16.08-, 19.43+, 25.10, 30.32+, 55.84-, 65.20+, 116.14 (q, J 288.2 Hz), 124.99+, 126.98+, 128.39+, 139.08, 156.40 (q, J 37.2 Hz); n
max: 1705 s, 1549 m, 1431 m, 1229 s, 1201 m, 1144 s, 698 m cm-1.
C: 16.08-, 19.43+, 25.10, 30.32+, 55.84-, 65.20+, 116.14 (q, J 288.2 Hz), 124.99+, 126.98+, 128.39+, 139.08, 156.40 (q, J 37.2 Hz); n
max: 1705 s, 1549 m, 1431 m, 1229 s, 1201 m, 1144 s, 698 m cm-1.
(1R, 4R, 5S)-1-Methyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid.
(1R, 4R, 5S)-1-Methyl-4-phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane (66.5 mg, 0.247 mmole,) was dissolved in carbon tetrachloride (0.8 ml) and acetonitrile (0.8 ml), water (1.2 ml), periodic acid (817 mg, 3.58 mmole, 14.5 eq.) and ruthenium trichloride hydrate (3.0 mg, 0.05 eq.) were added. The reaction mixture was vigorously stirred for 3 h at reflux (75 o
C), cooled to room temperature, and ether (5 ml) was added. The organic solution was separated and the water layer was washed with ether (2 x 2 ml). The combined ether layers were dried with magnesium sulfate and solvent was removed in vacuo to give (1R, 4R, 5S)-1-methyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid (32) (54.5 mg, 0.230 mmole, 93 %) as mixture of two rotamers (5:1), [ ]D20 +96.1o
(c 0.960, CHCl3) which showed
]D20 +96.1o
(c 0.960, CHCl3) which showed  H: 0.76 (1H, dd, J 8.0, 5.6 Hz), 0.98 (1H, dd, J 5.6, 4.0 Hz), 1.34 (3H, s), 1.74 (1H, ddd, J 8.0, 5.3, 4.0 Hz), 3.68 (1H, d, J 10.3 Hz), 3.95 (1H, d, J 10.3 Hz), 4.66 (1H, d, J 5.3 Hz), 9.59 (1H, s);
H: 0.76 (1H, dd, J 8.0, 5.6 Hz), 0.98 (1H, dd, J 5.6, 4.0 Hz), 1.34 (3H, s), 1.74 (1H, ddd, J 8.0, 5.3, 4.0 Hz), 3.68 (1H, d, J 10.3 Hz), 3.95 (1H, d, J 10.3 Hz), 4.66 (1H, d, J 5.3 Hz), 9.59 (1H, s);  C: 15.35-, 18.92+, 24.72-, 25.36, 54.77-, 62.35+, 115.83 (q, J 287.1 Hz), 156.45 (q, J 37.2 Hz), 173.60; n
max (CCl4): 3200 br.s, 2958 m, 2931 m, 1726 s, 1708 s, 1450 s, 1264 m, 1229 s, 1176 s, 1150 s, 761 m, 721 m cm-1; (m\z, %): 237, 5 (M+); 193, 14; 192, 100; 164, 10; 122, 8; 84, 39. In addition to the signals for the major rotamer given above, the minor rotamer showed
C: 15.35-, 18.92+, 24.72-, 25.36, 54.77-, 62.35+, 115.83 (q, J 287.1 Hz), 156.45 (q, J 37.2 Hz), 173.60; n
max (CCl4): 3200 br.s, 2958 m, 2931 m, 1726 s, 1708 s, 1450 s, 1264 m, 1229 s, 1176 s, 1150 s, 761 m, 721 m cm-1; (m\z, %): 237, 5 (M+); 193, 14; 192, 100; 164, 10; 122, 8; 84, 39. In addition to the signals for the major rotamer given above, the minor rotamer showed  H: 1.35 (3H, s), 5.55 (1H, d, J 5.1 Hz).
H: 1.35 (3H, s), 5.55 (1H, d, J 5.1 Hz).
(1R, 4R, 5S)-1-Methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid.
(1R, 4R, 5S)-1-Methyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid (35.5 mg, 0.149 mmole) was dissolved in a 1M solution of potassium hydroxide in methanol (0.5 ml) and this was stirred for 2 h. Solvent was removed in vacuo and the residue was dissolved in water, neutralized with 2M hydrochloric acid and desalted on a column of Dowex 50W - X8 resin in the hydrogen form. The amino acid was eluted from the column with 5M ammonia. After evaporation in vacuo and drying over phosphorus pentoxide (1R, 4R, 5S)-1-methyl-3-azabicyclo[3.1.0]hexane-4-carboxylic acid (33) was obtained (19.1 mg, 0.135 mmole, 91 %), m.p. 234 - 237 o
C (decomp.), [ ]D20 +107o
(c 0.63, H2O) which showed
]D20 +107o
(c 0.63, H2O) which showed  H (D2O): 0.89 - 0.99 (2H, m), 1.53 (3H, s), 2.04 (1H, ddd, J 8.3, 4.4, 4.0 Hz), 3.56 (1H, d, J 11.4 Hz), 3.72 (1H, d, J 11.4 Hz), 4.59 (1H, d, J 4.4 Hz);
H (D2O): 0.89 - 0.99 (2H, m), 1.53 (3H, s), 2.04 (1H, ddd, J 8.3, 4.4, 4.0 Hz), 3.56 (1H, d, J 11.4 Hz), 3.72 (1H, d, J 11.4 Hz), 4.59 (1H, d, J 4.4 Hz);  C (D2O): 13.41-, 19.42+, 26.04, 27.39+, 53.99-, 65.61+, 176.08; n
max(KBr): 3421 br. w, 2940 w, 2300 - 2800 w, 1599 s, 1458 w, 1393 s, 1372 s, 1341 s, 702 m cm-1.
C (D2O): 13.41-, 19.42+, 26.04, 27.39+, 53.99-, 65.61+, 176.08; n
max(KBr): 3421 br. w, 2940 w, 2300 - 2800 w, 1599 s, 1458 w, 1393 s, 1372 s, 1341 s, 702 m cm-1.
(1R, 4R, 5S)-N-t-Butoxycarbonyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane.
Crude (1R, 4R, 5S)-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (425 mg, >80 % purity by glc and PMR) was dissolved in THF (15 ml) and potassium carbonate (0.53 g, 5.0 mmole) and t-butoxycarbonylanhydride (0.61 g, 2.8 mmole) were added. The mixture was vigorously stirred for 3 h, filtered and THF was removed. Aq. sodium hydroxide (0.08 g, 2 mmole) in ethanol (10 ml) was added to the residue and the mixture was stirred for 1 h. The solvent was removed and the crude product was purified by passing through a column of silica (the faster the column, the higher the yield) eluting with petrol and ether (9:1) to yield (1R, 4R, 5S)-N-t-butoxycarbonyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (34) (391 mg, 1.43 mmole, 61 %), [ ]D20 +153.3o
(c 1.002, CHCl3) which showed
]D20 +153.3o
(c 1.002, CHCl3) which showed  H: 0.38 (1H, dd, J 7.9, 5.4 Hz), 0.63 (1H, br. s), 1.12 (9H, br. s), 1.28 (3H, s), 1.56 (1H, ddd, J 7.9, 4.9, 4.3 Hz), 3.49 (1H, d, J 10.4 Hz), 3.74 (1H, d, J 10.4 Hz), 4.98 (1H, d, J 4.3 Hz), 7.12 - 7.32 (5H, m);
H: 0.38 (1H, dd, J 7.9, 5.4 Hz), 0.63 (1H, br. s), 1.12 (9H, br. s), 1.28 (3H, s), 1.56 (1H, ddd, J 7.9, 4.9, 4.3 Hz), 3.49 (1H, d, J 10.4 Hz), 3.74 (1H, d, J 10.4 Hz), 4.98 (1H, d, J 4.3 Hz), 7.12 - 7.32 (5H, m);  C: 15.09-, 19.52+, 23.29, 27.98+, 31.86+, 56.18-, 63.64+, 79.33, 125.50+, 126.28+, 127.86+, 155.47; n
max: 3064 m, 3030 m, 2973 s, 2928 s, 2870 s, 1694 s, 1496 m, 1479 m, 1454 s, 1372 s, 1348 s, 1326 m,1282 m, 1177 s, 1128 s, 1101 m, 1076 m, 1029 m, 887 m, 760 m, 750 m, 701 s cm-1.
C: 15.09-, 19.52+, 23.29, 27.98+, 31.86+, 56.18-, 63.64+, 79.33, 125.50+, 126.28+, 127.86+, 155.47; n
max: 3064 m, 3030 m, 2973 s, 2928 s, 2870 s, 1694 s, 1496 m, 1479 m, 1454 s, 1372 s, 1348 s, 1326 m,1282 m, 1177 s, 1128 s, 1101 m, 1076 m, 1029 m, 887 m, 760 m, 750 m, 701 s cm-1.
(1R, 2S)-2-Benzoyl-1-methylcyclopropane-1-carboxylic acid.
(1R, 4R, 5S)-N-t-Butoxycarbonyl-1-methyl-4-phenyl-3-azabicyclo[3.1.0]hexane (12.3 mg, 0.045 mmole) was dissolved in carbon tetrachloride (0.2 ml) and acetonitrile (0.2 ml), water (0.3 ml), periodic acid (146 mg, 0.64 mmole, 14.2 eq.), and ruthenium trichloride hydrate (0.5 mg, 0.05 eq.) were added. The mixture was vigorously stirred for 3 h at room temperature, then ether (0.5 ml) was added. The black organic solution was separated and the water layer was washed with ether (2 x 0.5 ml). The ether solutions were combined and extracted with 0.1M sodium hydroxide (2 ml). Hydrochloric acid (0.5 ml, 1M) was added to the water layer and the solution was extracted with ether (2 x 5 ml). The ethereal solutions were combined, dried with magnesium sulfate and solvent was removed in vacuo to give (1R, 2S)-2-benzoyl-1-methylcyclopropane-1-carboxylic acid (35) (4.1 mg, 0.020 mmole, 44 %) which showed  H: 1.28 (1H, dd, J 8.3, 4.8 Hz), 1.54 (3H, s), 2.08 (1H, dd, J 7.0, 4.8 Hz), 2.60 (1H, dd, J 8.3, 7.0 Hz), 7.40 - 7.94 (5H, m); n
max (CDCl3): 3420 s br., 2924 s, 1775 s, 1709 s, 1467 m, 1369 m, 1158 m cm-1; (m/z (Me-ester), %): 218, 3 (M+); 187, 16; 186, 51; 159, 27; 158, 84; 105, 100; 91, 11; 77, 81.
H: 1.28 (1H, dd, J 8.3, 4.8 Hz), 1.54 (3H, s), 2.08 (1H, dd, J 7.0, 4.8 Hz), 2.60 (1H, dd, J 8.3, 7.0 Hz), 7.40 - 7.94 (5H, m); n
max (CDCl3): 3420 s br., 2924 s, 1775 s, 1709 s, 1467 m, 1369 m, 1158 m cm-1; (m/z (Me-ester), %): 218, 3 (M+); 187, 16; 186, 51; 159, 27; 158, 84; 105, 100; 91, 11; 77, 81.
(1R, 2S, 5S)-N-Benzyl-2-phenyl-3-azabicyclo[3.1.0]hexane.
A 1.5M solution of methyllithium in ether (10.9 ml, 16.4 mmole) was added to (R)-(+)-N,N-dibenzyl-2,2-dibromocyclopropylmethylamine (6.40 g, 15.6 mmole) in dry ether (150 ml) under nitrogen at -90 o
C over 5 min. The solution was stirred for 30 min at -90 o
C then warmed to 0 o
C for 30 min and water (20 ml) was added. The water layer was extracted with ether (50 ml). After drying the combined organic layers and removing the ether, the mixture was purified by chromatography over silica (ether - petrol, 1:49 to 1:4)) to give (1R, 2S, 5S)-N-benzyl-2-phenyl-3-azabicyclo[3.1.0]hexane (36) (3.26 g, 13.1 mmole, 84 %), (Found: M+, 249.1518. Calculated for C18H19N: 249.1518), Rf 0.67 (petrol - ether, 5:95), [ ]D20-145.5o
(c 1.078, CHCl3) which showed
]D20-145.5o
(c 1.078, CHCl3) which showed  H: 0.31 (1H, ddd, J 7.7, 7.7, 4.5 Hz), 1.11 (1H, ddd, J 4.5, 3.7, 3.7 Hz), 1.47 (1H, dddd, J 7.7, 6.8, 3.8, 3.7 Hz), 1.69 (1H, dddd, J 7.7, 6.8, 3.8, 3.7 Hz), 2.52 (1H, dd, J 8.8, 3.8 Hz), 3.16 (1H, d, J 8.8 Hz), 3.18 (1H, d, J 13.4 Hz), 3.82 (1H, d, J 3.8 Hz), 3.88 (1H, d, J 13.4 Hz), 7.26 - 7.60 (10H, m);
H: 0.31 (1H, ddd, J 7.7, 7.7, 4.5 Hz), 1.11 (1H, ddd, J 4.5, 3.7, 3.7 Hz), 1.47 (1H, dddd, J 7.7, 6.8, 3.8, 3.7 Hz), 1.69 (1H, dddd, J 7.7, 6.8, 3.8, 3.7 Hz), 2.52 (1H, dd, J 8.8, 3.8 Hz), 3.16 (1H, d, J 8.8 Hz), 3.18 (1H, d, J 13.4 Hz), 3.82 (1H, d, J 3.8 Hz), 3.88 (1H, d, J 13.4 Hz), 7.26 - 7.60 (10H, m);  C: 4.78-, 14.73+, 23.96+, 54.25-, 56.79-, 68.17+, 126.71+, 127.14+, 127.55+, 128.11+, 128.43+, 128.51+, 139.65, 142.27; n
max: 3060 m, 3001 m, 2786 s, 1493 s, 1453 s, 1361 s, 1344 m, 1280 m, 1025 s, 747 s, 699 s cm-1, (m/z, %): 250, 100; 249, 95 (M+); 248, 64; 173, 22; 172, 28; 159, 29; 158, 37; 92, 21; 91, 39.
C: 4.78-, 14.73+, 23.96+, 54.25-, 56.79-, 68.17+, 126.71+, 127.14+, 127.55+, 128.11+, 128.43+, 128.51+, 139.65, 142.27; n
max: 3060 m, 3001 m, 2786 s, 1493 s, 1453 s, 1361 s, 1344 m, 1280 m, 1025 s, 747 s, 699 s cm-1, (m/z, %): 250, 100; 249, 95 (M+); 248, 64; 173, 22; 172, 28; 159, 29; 158, 37; 92, 21; 91, 39.
Also were isolated N,N-dibenzyl-N-buta-2,3-dienylamine (38) (176 mg, 0.71 mmole, 4.5 %), (Found: M+, 249.1518. Calculated for C18H19N: 249.1518), Rf 0.51 (petrol - ether, 5:95) which showed  H: 3.17 (2H, dt, J 7.1, 2.5 Hz), 3.67 (4H, s), 4.74 (2H, dt, J 6.6, 2.5 Hz), 5.23 (1H, tt, J 7.1, 6.6 Hz), 7.24 - 7.54 (10H, m);
H: 3.17 (2H, dt, J 7.1, 2.5 Hz), 3.67 (4H, s), 4.74 (2H, dt, J 6.6, 2.5 Hz), 5.23 (1H, tt, J 7.1, 6.6 Hz), 7.24 - 7.54 (10H, m);  C: 51.71-, 57.42-, 74.83-, 86.45+, 126.99+, 128.28+, 128.99+, 139.35, 209.67; n
max: 3383 br. m, 3061 s, 3026 s, 2924 s, 2797 s, 1953 s, 1494 s, 1454 s, 1364 s, 1121 s, 738 s, 697 s cm-1; (m/z, %): 250, 6; 249, 7 (M+); 248, 5; 211, 54; 210, 59; 118, 7; 92, 96; 91, 100, 66, 25; 65, 62; (1R, 2R, 5S)-N-benzyl-2-phenyl-3-azabicyclo[3.1.0]hexane (37) (71 mg, 0.28 mmole, 1.8 %), (Found: M+, 249.1518. Calculated for C18H19N: 249.1518), Rf 0.41 (petrol - ether, 5:95), [
C: 51.71-, 57.42-, 74.83-, 86.45+, 126.99+, 128.28+, 128.99+, 139.35, 209.67; n
max: 3383 br. m, 3061 s, 3026 s, 2924 s, 2797 s, 1953 s, 1494 s, 1454 s, 1364 s, 1121 s, 738 s, 697 s cm-1; (m/z, %): 250, 6; 249, 7 (M+); 248, 5; 211, 54; 210, 59; 118, 7; 92, 96; 91, 100, 66, 25; 65, 62; (1R, 2R, 5S)-N-benzyl-2-phenyl-3-azabicyclo[3.1.0]hexane (37) (71 mg, 0.28 mmole, 1.8 %), (Found: M+, 249.1518. Calculated for C18H19N: 249.1518), Rf 0.41 (petrol - ether, 5:95), [ ]D20 +66.5o
(c 1.192, CHCl3) which showed
]D20 +66.5o
(c 1.192, CHCl3) which showed  H: 0.73 (1H, ddd, J 7.8, 7.8, 4.1 Hz), 1.01 (1H, ddd, J 4.1, 3.2, 3.2 Hz), 1.58 (1H, ddd, J 7.8, 7.3, 3.2 Hz), 1.69 (1H, dddd, J 7.8, 7.3, 3.2, 3.1 Hz), 2.94 (1H, d, J 9.0 Hz), 2.97 (1H, dd, J 9.0, 3.1 Hz), 3.33 (1H, d, J 14.0 Hz), 3.52 (1H, d, J 14.0 Hz), 4.09 (1H, s), 7.25 - 7.49 (10H, m);
H: 0.73 (1H, ddd, J 7.8, 7.8, 4.1 Hz), 1.01 (1H, ddd, J 4.1, 3.2, 3.2 Hz), 1.58 (1H, ddd, J 7.8, 7.3, 3.2 Hz), 1.69 (1H, dddd, J 7.8, 7.3, 3.2, 3.1 Hz), 2.94 (1H, d, J 9.0 Hz), 2.97 (1H, dd, J 9.0, 3.1 Hz), 3.33 (1H, d, J 14.0 Hz), 3.52 (1H, d, J 14.0 Hz), 4.09 (1H, s), 7.25 - 7.49 (10H, m);  C: 9.60-, 16.48+, 22.74+, 52.34-, 54.07-, 66.46+, 140.39, 142.24; n
max: 3061 m, 3027 s, 2794 s, 1493 s, 1453 s, 1366 m, 1172 m, 750 s, 700 s cm-1; (m/z, %): 250, 17; 249, 51 (M+); 248, 18; 197, 20; 172, 73; 158, 31; 106, 13; 91, 100; (1R, 2R)-N-(2-bromo-2-methylcyclopropylmethyl)-N,N-dibenzylamine (39) (86 mg, 0.25 mmole, 1.6 %) as colourless, viscous oil (Found: M+, 343.0936. Calculated for C19H22NBr: 343.0936), Rf 0.38 (petrol - ether, 5:95), [
C: 9.60-, 16.48+, 22.74+, 52.34-, 54.07-, 66.46+, 140.39, 142.24; n
max: 3061 m, 3027 s, 2794 s, 1493 s, 1453 s, 1366 m, 1172 m, 750 s, 700 s cm-1; (m/z, %): 250, 17; 249, 51 (M+); 248, 18; 197, 20; 172, 73; 158, 31; 106, 13; 91, 100; (1R, 2R)-N-(2-bromo-2-methylcyclopropylmethyl)-N,N-dibenzylamine (39) (86 mg, 0.25 mmole, 1.6 %) as colourless, viscous oil (Found: M+, 343.0936. Calculated for C19H22NBr: 343.0936), Rf 0.38 (petrol - ether, 5:95), [ ]D20 (c 1.224, CHCl3) which showed
]D20 (c 1.224, CHCl3) which showed  H: 0.80 (1H, dd, J 6.4, 4.9 Hz), 0.91 (1H, dddd, J 9.0, 6.4, 5.5, 5.3 Hz), 0.99 (1H, dd, J 9.0, 4.9 Hz), 1.77 (3H, s), 2.74 (1H, dd, J 13.2, 5.3 Hz), 2.84 (1H, dd, J 13.2, 5.5 Hz), 3.69 (2H, d, J 13.7 Hz), 3.80 (2H, d, J 13.7 Hz), 7.23 - 7.48 (10H, m);
H: 0.80 (1H, dd, J 6.4, 4.9 Hz), 0.91 (1H, dddd, J 9.0, 6.4, 5.5, 5.3 Hz), 0.99 (1H, dd, J 9.0, 4.9 Hz), 1.77 (3H, s), 2.74 (1H, dd, J 13.2, 5.3 Hz), 2.84 (1H, dd, J 13.2, 5.5 Hz), 3.69 (2H, d, J 13.7 Hz), 3.80 (2H, d, J 13.7 Hz), 7.23 - 7.48 (10H, m);  C: 22.22-, 23.57+, 30.74+, 37.68, 56.92-, 58.36-, 126.86+, 128.23+, 128.79+, 139.98; n
max: 3062 m, 3026 s, 2961 m, 2922 m, 2795 s, 1494 s, 1452 s, 1370 m, 1172 s, 1128 m, 1074 m, 1028m cm-1; (m/z, %): 346, 5; 345, 17; 344, 12; 343, 14 (M+); 342, 5; 210, 62; 91, 100; one isomer of bicyclopropylidene (41) (72 mg, 0.145 mmole, 1.8 %), Rf 0.17 (petrol - ether, 5:95), [
C: 22.22-, 23.57+, 30.74+, 37.68, 56.92-, 58.36-, 126.86+, 128.23+, 128.79+, 139.98; n
max: 3062 m, 3026 s, 2961 m, 2922 m, 2795 s, 1494 s, 1452 s, 1370 m, 1172 s, 1128 m, 1074 m, 1028m cm-1; (m/z, %): 346, 5; 345, 17; 344, 12; 343, 14 (M+); 342, 5; 210, 62; 91, 100; one isomer of bicyclopropylidene (41) (72 mg, 0.145 mmole, 1.8 %), Rf 0.17 (petrol - ether, 5:95), [ ]D20 +70.2o
(c 1.224, CHCl3) which showed
]D20 +70.2o
(c 1.224, CHCl3) which showed  H: 0.89 (1H, m), 1.34 (1H, m), 1.77 (1H, br. s), 2.37 (1H, dd, J 13.1, 7.2 Hz), 2.74 (1H, dd, J 13.1, 5.7 Hz), 3.63 (2H, d, J 13.8 Hz), 3.78 (2H, d, J 13.8 Hz), 7.21 - 7.42 (10H, m);
H: 0.89 (1H, m), 1.34 (1H, m), 1.77 (1H, br. s), 2.37 (1H, dd, J 13.1, 7.2 Hz), 2.74 (1H, dd, J 13.1, 5.7 Hz), 3.63 (2H, d, J 13.8 Hz), 3.78 (2H, d, J 13.8 Hz), 7.21 - 7.42 (10H, m);  C: 8.60-, 13.93+, 56.48-, 57.95-, 115.12, 126.79+, 128.22+, 128.72+, 140.03; n
max: 3061 m, 3027 s, 2969 m, 2794 s, 1494 s, 1453 s, 1367 s, 1126 m, 1074 m, 734 s, 698 s cm-1; a second isomer of bicyclopropylidene (40) (143 mg, 0.29 mmole, 3.6 %), Rf 0.13 (petrol - ether, 5:95), [
C: 8.60-, 13.93+, 56.48-, 57.95-, 115.12, 126.79+, 128.22+, 128.72+, 140.03; n
max: 3061 m, 3027 s, 2969 m, 2794 s, 1494 s, 1453 s, 1367 s, 1126 m, 1074 m, 734 s, 698 s cm-1; a second isomer of bicyclopropylidene (40) (143 mg, 0.29 mmole, 3.6 %), Rf 0.13 (petrol - ether, 5:95), [ ]D20 +23.6o
(c 1.010, CHCl3) which showed
]D20 +23.6o
(c 1.010, CHCl3) which showed  H: 0.96 (1H, m), 1.39 (1H, m), 1.79 (1H, br. s), 2.54 (1H, dd, J 13.3, 6.2 Hz), 2.67 (1H, dd, J 13.3, 6.5 Hz), 3.63 (2H, d, J 13.8 Hz), 3.80 (2H, d, J 13.8 Hz), 7.26 - 7.49 (10H, m);
H: 0.96 (1H, m), 1.39 (1H, m), 1.79 (1H, br. s), 2.54 (1H, dd, J 13.3, 6.2 Hz), 2.67 (1H, dd, J 13.3, 6.5 Hz), 3.63 (2H, d, J 13.8 Hz), 3.80 (2H, d, J 13.8 Hz), 7.26 - 7.49 (10H, m);  C: 8.80-, 13.57+, 56.75-, 57.94-, 115.34, 126.76+, 128.19+, 128.75+, 140.09; n
max: 3062 m, 3027 s, 2957 s, 2627 s, 2794 s, 1494 s, 1454 s, 1368 s, 1125 m, 1074 m, 1028 m, 735 s, 697 s cm-1; (m/z, %): 500, 100; 499, 63 (M+); 423, 9; 408, 17; 407, 11; 210, 13; 107, 9.
C: 8.80-, 13.57+, 56.75-, 57.94-, 115.34, 126.76+, 128.19+, 128.75+, 140.09; n
max: 3062 m, 3027 s, 2957 s, 2627 s, 2794 s, 1494 s, 1454 s, 1368 s, 1125 m, 1074 m, 1028 m, 735 s, 697 s cm-1; (m/z, %): 500, 100; 499, 63 (M+); 423, 9; 408, 17; 407, 11; 210, 13; 107, 9.
(1R, 2S, 5S)-2-Phenyl-3-azabicyclo[3.1.0]hexane.
(1R, 2S, 5S)-N-Benzyl-2-phenyl-3-azabicyclo[3.1.0]hexane (2.59 g, 10.4 mmole) was dissolved in ethanol (50 ml) and 3M hydrochloric acid (6.9 ml, 20.8 mmole) and 5 % Pd/C (1.3 g) were added. After stirring for 2 h under hydrogen, the mixture was filtered, ethanol was evaporated and 2 % aq. sodium hydroxide (50 ml, 25 mmole) was added. The amine was extracted with ether (3 x 50 ml), the organic solution was dried and solvent was removed to give (1R, 2S, 5S)-2-phenyl-3-azabicyclo[3.1.0]hexane (1.61 g, 10.1 mmole, 97 %), (Found: M+, 159.1048. Calculated for C11H13N: 159.1048), [ ]D20-117.9o
(c 1.062, CHCl3) which showed
]D20-117.9o
(c 1.062, CHCl3) which showed  H: 0.37-0.51 (2H, m), 1.44 (1H, m), 1.60 (1H, m), 2.06 (1H, s), 3.01 (1H, d, J 11.1 Hz), 3.05 (1H, dd, J 11.1, 2.9 Hz), 4.30 (1H, d, J 3.2 Hz), 7.15 - 7.43 (5H, m);
H: 0.37-0.51 (2H, m), 1.44 (1H, m), 1.60 (1H, m), 2.06 (1H, s), 3.01 (1H, d, J 11.1 Hz), 3.05 (1H, dd, J 11.1, 2.9 Hz), 4.30 (1H, d, J 3.2 Hz), 7.15 - 7.43 (5H, m);  C: 3.58-, 16.27+, 20.89+, 49.23-, 62.53+, 127.08+, 127.28+, 128.36+, 141.69; n
max: 3317 br. s, 3061 m, 3031 s, 2925 s, 2864 s, 1495 m, 1451 s, 1089 m, 1032 m, 927 m, 745 s, 700 s cm-1; (m/z, %): 159, 100 (M+); 158, 90; 144, 12; 129, 16; 105, 23; 104, 32; 92, 15; 91, 28; 83, 27; 82, 42; 54, 17.
C: 3.58-, 16.27+, 20.89+, 49.23-, 62.53+, 127.08+, 127.28+, 128.36+, 141.69; n
max: 3317 br. s, 3061 m, 3031 s, 2925 s, 2864 s, 1495 m, 1451 s, 1089 m, 1032 m, 927 m, 745 s, 700 s cm-1; (m/z, %): 159, 100 (M+); 158, 90; 144, 12; 129, 16; 105, 23; 104, 32; 92, 15; 91, 28; 83, 27; 82, 42; 54, 17.
(1R, 2S, 5S)-2-Phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane.
(1R, 2S, 5S)-2-Phenyl-3-azabicyclo[3.1.0]hexane (1.53 g, 9.61 mmole) was dissolved in dichloromethane (30 ml) and trifuoroacetic anhydride (10 ml, 50 mmole) was added. The solution was stirred for 2 h, then the volatiles were removed in vacuo and the residue was dissolved in ether (50 ml) and this solution was extracted with phosphate buffer (2 x 30 ml, pH 6.88). The organic solution was dried and, after removing the solvent, (1R, 2S, 5S)-2-phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane (42) (2.44 g, 9.56 mmole, 99 %) was obtained, m.p. 94 - 97 o
C, [ ]D20-170.4o
(c 1.004, CHCl3), 93.3 % e.e. by chiral g.l.c. (155 o
C). This crude product was recrystallysed from hexane - ethyl acetate (20 ml, 17:3) and the mother liquor after this was recrystallysed as well to yield (1R, 2S, 5S)-2-phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane (42) (2.06 g, 8.07 mmole, 84 %), (Found: C 61.03, H 4.74, N 5.49. Calculated for C13H12NOF3: C 61.18, H 4.65, N 5.66), m.p. 107 - 108 o
C, [
]D20-170.4o
(c 1.004, CHCl3), 93.3 % e.e. by chiral g.l.c. (155 o
C). This crude product was recrystallysed from hexane - ethyl acetate (20 ml, 17:3) and the mother liquor after this was recrystallysed as well to yield (1R, 2S, 5S)-2-phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane (42) (2.06 g, 8.07 mmole, 84 %), (Found: C 61.03, H 4.74, N 5.49. Calculated for C13H12NOF3: C 61.18, H 4.65, N 5.66), m.p. 107 - 108 o
C, [ ]D20-192.7o
(c 0.999, CHCl3), 100.0 % e.e. by chiral g.l.c. (155 o
C) which showed
]D20-192.7o
(c 0.999, CHCl3), 100.0 % e.e. by chiral g.l.c. (155 o
C) which showed  H: 0.46 (1H, ddd, J 6.1, 4.1, 4.1 Hz), 0.68 (1H, ddd, J 8.1, 7.9, 6.1 Hz), 1.87 (1H, dddd, J 7.9, 7.6, 5.0, 4.1 Hz), 2.03 (1H, dddd, J 8.1, 7.6, 5.5, 4.1 Hz), 3.99 (1H, d, J 10.7 Hz), 4.11 (1H, dd, J 10.7, 5.0 Hz), 5.38 (1H, d, J 5.5 Hz), 7.09 - 7.40 (5H, m);
H: 0.46 (1H, ddd, J 6.1, 4.1, 4.1 Hz), 0.68 (1H, ddd, J 8.1, 7.9, 6.1 Hz), 1.87 (1H, dddd, J 7.9, 7.6, 5.0, 4.1 Hz), 2.03 (1H, dddd, J 8.1, 7.6, 5.5, 4.1 Hz), 3.99 (1H, d, J 10.7 Hz), 4.11 (1H, dd, J 10.7, 5.0 Hz), 5.38 (1H, d, J 5.5 Hz), 7.09 - 7.40 (5H, m);  C: 10.10-, 17.81+, 23.85+, 51.09 (-, q, J 3.8 Hz), 64.70+, 116.20 (q, J 288.2 Hz), 125.08+, 127.02+, 128.46+, 139.02, 156.49 (q, J 36.9 Hz); n
max (CCl4): 2900 m, 1694 s, 1495 m, 1453 m, 1370 m, 1345 m, 1224 s, 1146 s, 1079 m, 836 m, 784 m, 749 m, 701 s cm-1; (m/z, %): 256, 42 (M+); 254, 100; 142, 45; 129, 29; 117, 31; 104, 33; 91, 21; 81, 19.
C: 10.10-, 17.81+, 23.85+, 51.09 (-, q, J 3.8 Hz), 64.70+, 116.20 (q, J 288.2 Hz), 125.08+, 127.02+, 128.46+, 139.02, 156.49 (q, J 36.9 Hz); n
max (CCl4): 2900 m, 1694 s, 1495 m, 1453 m, 1370 m, 1345 m, 1224 s, 1146 s, 1079 m, 836 m, 784 m, 749 m, 701 s cm-1; (m/z, %): 256, 42 (M+); 254, 100; 142, 45; 129, 29; 117, 31; 104, 33; 91, 21; 81, 19.
(1R, 2S, 5S)-N-Trifluoroacetyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid.
(1R, 2S, 5S)-2-Phenyl-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane (1.68 g, 6.58 mmole,) was dissolved in carbon tetrachloride (16 ml) and acetonitrile (16 ml), water (24 ml) and periodic acid (21.75 g, 95.4 mmole, 14.5 eq.) were added. After addition of ruthenium trichloride hydrate (74 mg, 0.05 eq.) the solution became black. The reaction mixture was warmed to 80 o
C with vigorous stirring when oxidation began (black colour echanged to yellowish and gas began to be evolved). After 2.75 h the reaction mixture became black again and after 1 h more it was cooled to room temperature and ether (50 ml) was added. The organic solution was separated and the water layer was washed with ether (2 x 50 ml). The combined ether layers were dried and the solvent was removed in vacuo to give (1R, 2S, 5S)-N-trifluoroacetyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid (43) (Found: M+, 223.0456. Calculated for C8H8NO3F3: 223.0456), (1.29 g, 5.78 mmole, 88 %) as a mixture of two rotamers (11:4), [ ]D20-116.5o
(c 0.960, CHCl3) which showed n
max : 3260 br.s, 1726 s, 1696 s, 1451 m, 1221 s, 1180 s, 1068 w cm-1; (m/z, %): 223, 2 (M+); 179, 12; 178, 100; 126, 7; 69, 31; major rotamer,
]D20-116.5o
(c 0.960, CHCl3) which showed n
max : 3260 br.s, 1726 s, 1696 s, 1451 m, 1221 s, 1180 s, 1068 w cm-1; (m/z, %): 223, 2 (M+); 179, 12; 178, 100; 126, 7; 69, 31; major rotamer,  H: 0.73 - 0.90 (2H, m), 1.82 - 2.19 (2H, m), 3.84 - 4.03 (2H, m), 4.63 (1H, d, J 5.5 Hz), 11.08 (1H, s);
H: 0.73 - 0.90 (2H, m), 1.82 - 2.19 (2H, m), 3.84 - 4.03 (2H, m), 4.63 (1H, d, J 5.5 Hz), 11.08 (1H, s);  C: 9.06-, 17.68+, 18.58+, 50.18 (-, q, J 3.4 Hz), 61.97+, 115.81 (q, J 286.9 Hz), 156.86 (q, J 38.1 Hz), 174.00; minor rotamer,
C: 9.06-, 17.68+, 18.58+, 50.18 (-, q, J 3.4 Hz), 61.97+, 115.81 (q, J 286.9 Hz), 156.86 (q, J 38.1 Hz), 174.00; minor rotamer,  H: 0.49 - 0.55 (1H, m), 0.73 - 0.90 (1H, m), 1.82 - 2.19 (2H, m), 3.84 - 4.03 (2H, m), 5.48 (1H, d, J 5.4 Hz), 11.08 (1H, s);
H: 0.49 - 0.55 (1H, m), 0.73 - 0.90 (1H, m), 1.82 - 2.19 (2H, m), 3.84 - 4.03 (2H, m), 5.48 (1H, d, J 5.4 Hz), 11.08 (1H, s);  C: 9.12-, 18.19+, 50.40-, 64.00+, 115.81 (q, J 286.9 Hz), 156.52 (q, J 38.4 Hz), 160.79.
C: 9.12-, 18.19+, 50.40-, 64.00+, 115.81 (q, J 286.9 Hz), 156.52 (q, J 38.4 Hz), 160.79.
(1R, 2S, 5S)-3-Azabicyclo[3.1.0]hexane-2-carboxylic acid.
(1R, 2S, 5S)-N-Trifluoroacetyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid (1.24 g, 5.56 mmole) was dissolved in a 1M solution of potassium hydroxide in methanol (15 ml) and this was stirred for 2 h. Then solvent was removed in vacuo and the residue was dissolved in water, neutralized with 2M hydrochloric acid and desalted on a column of Dowex 50W - X8 resin in the hydrogen form. The amino acid was eluted from the column with 5M ammonia. After evaporation in vacuo, drying over phosphorus pentoxide and recrystallising from ethanol - acetone, (1R, 2S, 5S)-3-azabicyclo[3.1.0]hexane-2-carboxylic acid (1) was obtained (557 mg, 4.38 mmole, 79 %), (Found: C 56.90, H 7.00, N 10.80. Required for C6H9NO2: C 56.68, H 7.35, N 11.02), m.p. 240 - 241 o
C (decomp.), (lit.9 m.p. 235 - 245 o
C), [ ]D20-141.8o
(c 0.776, H2O), (lit.1 [
]D20-141.8o
(c 0.776, H2O), (lit.1 [ ]D29-132o
; lit.9 [
]D29-132o
; lit.9 [ ]D20-144o
) which showed
]D20-144o
) which showed  H (D2O): 0.48 (1H, ddd, J 6.5, 4.2, 4.2 Hz), 0.77 (1H, ddd, J 8.2, 8.2, 6.5 Hz), 1.83 (1H, m), 2.05 (1H, dddd, J 8.2, 6.7, 4.6, 4.2 Hz), 3.48 (2H, d, J 2.0 Hz), 4.28 (1H, d, J 4.6 Hz);
H (D2O): 0.48 (1H, ddd, J 6.5, 4.2, 4.2 Hz), 0.77 (1H, ddd, J 8.2, 8.2, 6.5 Hz), 1.83 (1H, m), 2.05 (1H, dddd, J 8.2, 6.7, 4.6, 4.2 Hz), 3.48 (2H, d, J 2.0 Hz), 4.28 (1H, d, J 4.6 Hz);  C (D2O): 7.05-, 18.26+, 21.24+, 50.30-, 65.23+, 175.93; n
max(KBr): 3432 br. w, 2980 w, 2300 - 2800 w, 1630 s, 1603 s, 1388 s, 1355 m, 706 m cm-1.
C (D2O): 7.05-, 18.26+, 21.24+, 50.30-, 65.23+, 175.93; n
max(KBr): 3432 br. w, 2980 w, 2300 - 2800 w, 1630 s, 1603 s, 1388 s, 1355 m, 706 m cm-1.
- Fowden, L.; Smith, A.; Millington, D.S.; Sheppard, R.C. Phytochemistry, 1969, 8, 437.
- Caveney, S.; Starratt, A.N. Nature (London), 1994, 372, 509.
- Starratt, A.N.; Caveney, S. Phytochemistry, 1995, 40, 479.
- Machackova, I.; Zmrhal, Z. Biol. Plant., 1983, 25 (5), 394.
- Kerr, M.W.; Ger.Offen 2641295 (1979); Kollmeyer, W.D.; Barrett, S.K.; Flint, D.H. Amer.Chem.Soc. Symp.Series, 1987, 355, 401.
- Patchett, A.A.;Taub, D.; Myvratt, M.J. S.African ZA 83 04457, U.S.Appl. 389,735.
- Rowland, I.; Tritham, H. J.Bact., 1975, 123, 871.
- Day, J.A.; Grayson, B.T. Brit.UK Pat.Appl. GB 2089337; Mason, R.F.; Wood, D.A. Eur.Pat.Appl. 9290; Brit.Appl. 78/38331; Kollmeyer, W.D., U.S. 413857; Wood, D.A.; Mason, R.F.; Day, J.A., Searle, R.J.G. Eur.Pat.Appl., 10799; Brit.Appl. 78/42282; U.S.Appl. 922408; Day, J.A.; Devlin, B.R.J.; Searle, R.J.G. Eur.Pat. Appl. 7652; Brit.Appl. 78/27978; U.S. 4254057; Scholes, G.; Boardman, F. U.S. 4262124; Brit.Appl. 79/64.
- Fujimoto, Y.; Irreverre, F.; Karie, J.M.; Karle, I.L.; Witkop, B. J.Amer.Chem.Soc., 1971, 93, 3471.
- Sagnard, I.; Sasaki, A.; Chiaroni, A.; Riche, C.; Potier, P. Tetrahedron Lett., 1995, 36, 3149.
- Baird, M.S.; Kaura, A.C. J. Chem. Soc., Chem. Comm., 1976, 356.
- Baird, M.S. J. Chem. Soc., Chem. Comm., 1971, 1145; Arct, J.; Skattebol, L. Acta Chem. Scand., 1982, 36, 593; Bolesov, I.G.; Tverezovsky, V.V.; Grishin, Yu.K. Zh.Org.Khim., 1997, 33 (1), 135-137.
- Al Dulayymi, A.R.; Al Dulayymi, J.R.; Baird, M.S; Gerrad, M.E.; Koza, E.; Harkins, S.M.; Roberts, E. Tetrahedron, 1996, 52, 3409.
- Baird, M.S.; Bolesov, I.G.; Tverezovsky, V.V.; Clegg, W. to be submitted.
- See eg., Miebach, T.; Wuster, H.; Brinker, U.H. J. Org. Chem., 1993, 58, 6520.
- Carlsen, Per H.G.; Katsuki, T.; Martin, V.S.; Sharpless, K.B. J. Org. Chem., 1981, 46, 3938.
- Backes, J.; Brinker, U. Methoden Der Organischen Chemie, Ed. M.Regitz, Houben Weyl, 19b, Part 1, 1989, 391.
- Baird, M.S.; Baxter, A.G.W. J. Chem. Soc., Perkin Transl. 1, 1979, 2317.
- Kleveland, K.; Skattebol, L. Acta Chem. Scand., 1977, B31, 463.
- Baird, M.S.; Baxter, A.G.W.; Devlin, B.R.J.; Searle, R.J.G. J. Chem. Soc., Chem. Comm., 1979, 210.
- Holm, K.H.; Lee, D.G.; Skattebol, L. Acta Chem. Scand., 1978, B32, 128.
- Labeish, N.N.; Kharicheva, E.M.; Mandel'shtam, T.V.; Kostikov, R.R. Zr.Org.Khim., 1978, 14, 878; Kharicheva, E.M.; Labeish, N.N.; Kostikov, R.R. Zr.Org.Khim., 1980, 16, 2513.